Video assisted thoracoscopic (VATS) left main bronchial sleeve resection with intracorporeal bronchial anastomosis using barbed sutures: a case report
Introduction
The main function of surgical knots in tissue function.is anchoring. Surgical knots reduce the tensile strength of all sutures by thinning and stretching the material. Knot-secured suture creates an uneven tension across the wound with the highest tension at the knots. Besides excessive tension, weakening of the suture, foreign body inflammatory reaction and delayed wound healing, tying of surgical knots introduces the potential of human error by inter-user variability (1). Furthermore, in minimally invasive surgeries, the ability to tie surgical knots is a challenge. The skills necessary to perform intra- or extracorporeal knot tying though can be achieved with practice it is difficult especially where exposure is suboptimal. Additionally, laparoscopically tied knots are often found to be weaker than those tied by hand or robotically (1). Therefore, in cases where knot tying and sewing is difficult, the use of barbed suture can be used with similar efficacy but with less time and cost (1-3).
Video assisted thoracoscopic (VATS) bronchial anastomosis are generally done extra-corporeally as in open surgery either interrupted or continuously due to simplicity. However continuous sutures can be complicated by the need for a bigger utility incision, entanglement of sutures, difficulty in maintaining suture tension and the need for extra surgical assistance. Interrupted sutures though easier to perform can be tedious taking a longer time (4,5) As intracorporeal suturing is easier to perform in robotic surgery as compared to VATS, the use of barbed sutures in robotic assisted sleeve resections is well reported (6-8). A totally intra-corporeal “DIY” bronchial anastomosis by VATS using a barbed suture is described.
The following article is presented in accordance with the CARE reporting checklist (available at https://jovs.amegroups.com/article/view/10.21037/jovs-20-120/rc).
Case presentation
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Patient selection, work up and preoperative preparation
Patient is a 35 years old Caucasian female who presented with symptoms of wheezing for 1 year. CT scan thorax showed a polypoidal growth arising from the proximal left main stem bronchus and extending to the junction of upper and lower lobe bronchus (Figure 1). Bronchoscopy and biopsy showed a low grade mucoepidermoid carcinoma (Figure 2). PET CT scan showed no evidence of distant metastases.
Equipment preference card
- 5 mm 30 degrees camera lens;
- 5 mm instruments including needle holder and Maryland dissector;
- Vessel loop/tape;
- 4-0 PDS barbed sutures (Medtronic, V-Lok sutures) ×2;
- Nerve Hook or 5 mm Hook dissector for tightening of sutures.
Procedure
Under general anaesthesia a right sided double lumen endobronchial tube was placed under bronchoscopic guidance as a left sided tube would be in the way of the surgical operative field.
Patient was placed in a right lateral decubitus position with flexion at the hips. The VATS was done totally endoscopically watching the TV monitor with the surgeon and camera man standing in front of the TV monitor and the second assistant on the opposite side (Figure 3). All incisions were placed in the anterior axillary line. 1×5 mm working port at 2nd ICS, 1×5 mm camera port at 4th ICS and 10 mm camera port at the 6th ICS were placed (Figure 4).
With the lung collapsed exploratory thoracoscopy was done. The lung was retracted posteriorly exposing anterior hilar structures. The mediastinal pleura over the left main pulmonary artery was divided and the main pa was mobilised circumferentially, taped, and retracted. Aorto-pulmonary lymph nodes stations 5 and 6 were systematically dissected with careful preservation of left recurrent laryngeal and vagus nerves. The anterior left main bronchus and lower trachea were mobilised. The lung was then retracted anteriorly towards the surgeon for exposure of the posterior hilum. The inferior ligament was released to decrease tension on the subsequent anastomosis. Stations 7 and 10 (subcarinal) lymph nodes were then dissected. The posterior aspect of left and right main bronchi, carina and lower trachea were all mobilised.
The tumour was noted to have extended extra-luminally through the posterior wall of bronchus. This was mobilised circumferentially from the surrounding tissues with adequate surgical margins. The bronchoscope was then introduced into the left main bronchus by the anaesthesiologist with the OR lights dimmed for surface marking the intraluminal extension of the tumour. This was done by cautery. A bronchotomy was done over the marked cauterized area. The tumour was then excised with adequate surgical margins. Additional distal and proximal bronchial margins were sent separately for pathological analysis. Once the surgical margins were confirmed to be clear of cancer, the distal and proximal bronchi were adequately mobilised for a tension free mucosa to mucosa anastomosis. To increase exposure under the aorta a stay suture was placed to elevate the aorta up away towards the chest-wall.
Anastomosis was started on the cartilaginous wall using a 4-0 PDS V-Loc barbed suture working from posterior to anterior. The suture is locked by passing the needle through the eye of the barbed suture. After a few posterior cartilaginous sutures, the lung was flipped posteriorly. With the lung and pulmonary artery retracted away, exposure was enhanced for completion of the rest of the cartilaginous part of anastomosis. The sutures were gently tightened using a hook dissector ensuring even distribution of tension on the anastomosis. Due to the self-locking nature of the suture, it does away the need for an assistant to maintain tension and prevent suture entanglement. Once the anterior cartilaginous anastomosis was completed, a new 4-0 PDS barbed suture was started on the distal membranous part again gain locking the suture through the suture eye. The sutures were placed loosely first at regular intervals and then subsequently tightened gently using a 5 mm hook dissector. This prevents tearing of the thin wall membranous portion of the bronchus by the braided suture. Once the anastomosis was completed it was tied to the cartilaginous suture with a few intracorporeal knots. Integrity of the anastomosis was check for air leak under 30 cm of water suction. There was a small air leak at the distal membranous and cartilaginous junction due to inadequate tightening of the suture. This was rectified by reinforcing it with a non-barbed 4-0 PDS suture. Repeat testing showed no further air leak. Intraoperative bronchoscopy showed a patent anastomosis. Operative time was 235 mins and blood loss was minimal. A single chest tube F20 was inserted and the wound was closed in layers (Video 1).
Post-operative management
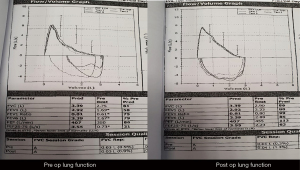
Chest tube was removed on 2nd post-operative day. Post op recovery was uneventful and patient was discharged on 4th post-operative day. Post-operative CXR was normal with improved lung function. (Figure 5). Post-operative bronchoscopy and PET CT follow up 1 year after surgery shows no tumour relapse (Figure 6). The whole process was shown in the timeline (Figure 7).
Tips and pitfalls
- Aim of surgery is to achieve an oncologically complete R0 resection. Meticulous dissection with clear surgical margins and systematic radical lymphadenectomy is mandatory. Lymphadenectomy additionally also increases better exposure of the hilar structures. Good hemostasis will ensure a clear surgical field during anastomosis. All bronchial margins must be subjected to intraoperative pathological analysis and confirmed to be negative for malignancy before anastomosis;
- The bronchial anastomosis must be well vascularised and tension free with mucosal-to-mucosal anastomosis. Tension on the anastomosis can be minimised by adequate mobilisation of the proximal and distal bronchus. Release procedures like division of the inferior ligament ligament should be routine. In long segment resections where tension can be excessive, circumferential intra-pericardial release of hilar structures should be done;
- Exposure is critical for accurate sewing especially on the left side due to limited surgical working space from crowding of the hilar structures, constant pulsating of the heart and great vessels and the deep origin of the left main bronchus under the aorta makes sewing challenging. Exposure can be improved by retraction of the arch of the aorta with a retraction suture anchored to the chest-wall, retraction of the pulmonary artery with a vascular loop or tape, adequate mobilisation of the carina and both main bronchi and lower trachea;
- VATS bronchoplasty needs proficiency in endoscopic sewing and tying. This is acquired by routine sewing whenever possible in standard Vats lobectomy followed by gaining gradual experience in performing simple and wedge bronchoplasties before embarking on complex sleeve bronchoplasties. Proficiency in both intra and extracorporeal sewing and tying is mandatory (4);
- General anastomosis preference is to do cartilaginous anastomosis before membranous so as to prevent tearing of thin wall membranous wall and for correction of size discrepancy between the two bronchial ends. Due to the braided nature of the barbed sutures care should be taken during placement and tightening of the sutures from tearing especially the membranous part of the bronchus. This can be minimised by placing the sutures first and then gently tightening with a nerve hook (4);
- All anastomosis should be routinely checked by intraoperative bronchoscopy for integrity. This allows for immediate remedial surgical corrections if necessary;
- There are many commercially available barbed sutures (Quill, V-Loc and STRATAFIX)). The same materials that are used to produce strands of smooth suture are reconfigured to produce suture with barbs on their surfaces. This is done by cutting into the shaft of the strand of smooth suture with a blade. The drawback is it reduces the tensile strength of the suture by weakening its core and narrowing its functional diameter. Due to this potential weakening, it is generally recommended to use a barbed suture one strength stronger than a traditional smooth suture to maintain the same tensile strength (1).
Conclusions
VATS bronchial anastomosis by extracorporeal sewing can be technically challenging due to suboptimal exposure, lack of experienced technical assistance, entanglement of sutures and adequate maintenance of proper suture line tension. This can be simplified by the adoption of intracorporeal sewing using a barbed suture.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The author has completed the CARE reporting checklist. Available at https://jovs.amegroups.com/article/view/10.21037/jovs-20-120/rc
Conflicts of Interest: The author has completed the ICMJE uniform disclosure form (available at https://jovs.amegroups.com/article/view/10.21037/jovs-20-120/coif). TA serves as an unpaid editorial board member of Journal of Visualized Surgery from June 2019 to May 2023. The author has no other conflicts of interest to declare.
Ethical Statement: The author is accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Greenberg JA, Goldman RH. Barbed suture: a review of the technology and clinical uses in obstetrics and gynecology. Rev Obstet Gynecol 2013;6:107-15. [PubMed]
- Nemecek E, Negrin L, Beran C, Nemecek R, Hollinsky C. The application of the V-Loc closure device for gastrointestinal sutures: a preliminary study. Surg Endosc 2013;27:3830-4. [Crossref] [PubMed]
- Nakagawa T, Chiba N, Ueda Y, et al. Clinical experience of sleeve lobectomy with bronchoplasty using a continuous absorbable barbed suture. Gen Thorac Cardiovasc Surg 2015;63:640-3. [Crossref] [PubMed]
- Agasthian T. Initial experience with video-assisted thoracoscopic bronchoplasty. Eur J Cardiothorac Surg 2013;44:616-23. [Crossref] [PubMed]
- Chakaramakkil MJ, Jim LY, Soon JL, et al. Continuous absorbable suture technique for tracheobronchial sleeve resections. Asian Cardiovasc Thorac Ann 2011;19:44-7. [Crossref] [PubMed]
- Egberts JH, Möller T, Hauser C, et al. Robotic assisted sleeve lobectomy with the use of barbed sutures. J Vis Surg 2018;4:117. [Crossref]
- Cosgun T, Kaba E, Ayalp K, et al. Bronchial sleeve anastomosis and primary closures with the da Vinci system: an advanced minimally invasive technique. Video-assist Thorac Surg 2017;2:49. [Crossref]
- Sarsam OM, Dunning J, Pochulu B, et al. Robot-assisted bronchoplasty using continuous barbed sutures. J Vis Surg 2018;4:3. [Crossref] [PubMed]
Cite this article as: Agasthian T. Video assisted thoracoscopic (VATS) left main bronchial sleeve resection with intracorporeal bronchial anastomosis using barbed sutures: a case report. J Vis Surg 2022;8:11.