Repair of a recurrent traumatic rectovaginal fistula using vaginal wall plication to reinforce a rectal wall advancement flap
Introduction
Although rectovaginal fistulas (RVFs) are not life threatening disorders, they are amongst the most challenging diseases for surgeons. Patients with RVFs typically complain of passing flatus or feces from the vagina. These symptoms may cause both physical discomfort and psychological distress. The most common cause of RVFs is obstetric trauma (1). One study demonstrated that concomitant fecal incontinence is seen in 48% of women with a RVF (2). Examination with digital rectal and vaginal exam, anoscopy, and speculum exam should confirm the diagnosis in most patients.
RVFs can be classified as simple or complex based on etiology, location, and size (3). Simple RVFs are low (rectal opening near dentate line and vaginal opening just inside the vaginal fourchette), small (<2.5 cm) and are typically caused by obstetric trauma or cryptoglandular infection. On the other hand, complex RVFs are high (vaginal opening at or near cervix), large, or associated with inflammatory bowel disease, radiation, cancer, or persistent despite attempts at prior repair. Rectal advancement flap repair (RAFR) is appropriate for most simple RVFs and can also be used on more complex RVFs though generally with less success (4).
We present a case of a 28-year-old female with a history of a traumatic cloaca repaired 12 years prior that was diagnosed with a recurrent RVF and underwent repair using a rectal wall advancement flap reinforced with posterior vaginal wall plication under protection of a loop ileostomy. This case is used to demonstrate a surgical technique that can be used to treat RVF in patients with significant tissue defects.
Methods
Patient selection and workup
The reported success rates of RAFR are highly variable ranging between 29% and 86% (2,3,5-16). This rate likely depends on the etiology of the fistula and the quality of the tissue used to repair the fistula. Better outcomes are achieved with primary procedure, simple fistulas, and fistulas caused by obstetric trauma. RAFR should be avoided in patients with active infection, inflammation, or neoplastic fistulas.
The etiology of the fistula should be sought after prior to performing a RAFR. Patients with traumatic fistulas should be evaluated for fecal incontinence and a concomitant sphincteroplasty should be considered when this symptom is present. If there are signs of an active infection, this should be controlled with drainage and possible seton placement prior to attempt at definitive repair. Also, fecal diversion can be considered with patients with a complex RVF. Patients with Crohn’s disease should be medically optimized prior to attempting repair. A history of pelvic radiation is a relative contraindication to RAFR; tissue transfer procedures should be considered in these patients.
Preoperative preparation
In our practice, patients typically undergo a full mechanical bowel preparation the day before surgery and preoperative antibiotics are administered within 30 min of initial incision. RAFR of a RVF is generally performed under general anesthesia. The patient is placed in the prone jackknife position with the buttocks taped apart. The anal canal and vagina are prepped with povidone-iodine solution.
Equipment preference card
- Anorectal tray;
- Headlight;
- Lone star retractor;
- Fine-tip electrocautery and forceps;
- 3-0 absorbable sutures;
- Suction device.
Procedure
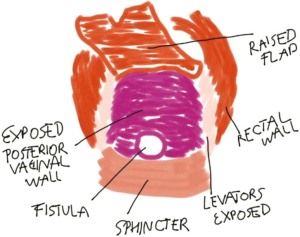
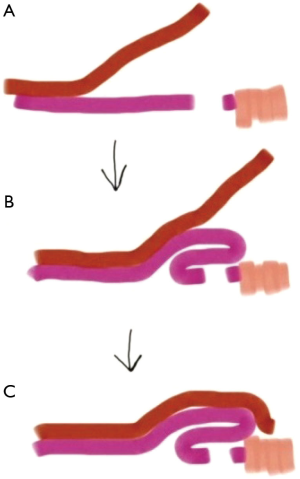
A digital rectal and vaginal exam should be performed along with anoscopy to confirm the presence of a fistula. A lone star retractor is then used to expose the rectal mucosa. The surgical technique is demonstrated in the video (Figure 1). Creation of a trapezoidal rectal advancement flap with the apex at the fistula opening is began by mobilizing the rectal mucosa just proximal and lateral to the fistula opening and carrying this incision in the proximal and lateral directions. Flap mobilization should continue 4–5 cm proximal to the fistula defect. The flap base should be at least twice the width of the apex to ensure adequate vascular supply. The dissection is carefully deepened exposing the levator muscles laterally. The flap is then retracted superiorly exposing the rectovaginal plane. The rectal flap is separated from the vagina through lateral to medial dissection along the rectovaginal plane. This approach avoids scar tissue and allows for easier identification of the correct plane. This maneuver is repeated along the opposite site of the flap. At the end of this dissection, the scar tissue at the fistula opening is incised connecting the two previously created planes in the rectovaginal septum. This dissection is carried proximally fully freeing the rectal flap from the posterior vaginal wall. The mobilization of the rectal flap should allow for a tension-free reconstruction. The anatomy visualized at this point is represented in Figure 2.

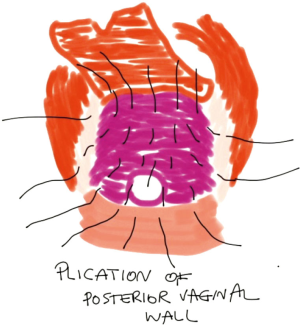
The fistula opening in the vaginal wall is identified and the scar tissue along the edges is debrided. The healthy edges of the vaginal wall can be loosely approximated using absorbable sutures. Alternatively, the vaginal opening can be left open for drainage. The posterior vaginal wall is dissected out as needed until enough pliable and redundant tissue is exposed to allow for plication over the fistula opening (Figure 3). This is done using 3-0 absorbable sutures (Figure 4). The proximal vaginal wall is sutured to the sphincter muscle complex during the plication process. In patients with a lax pelvic floor, a levatorplasty can be performed to reinforce the vaginal repair. At the completion of the vaginal wall plication, the rectal flap should be brought distally allowing for tension-free repair. The apex of the rectal flap can be trimmed and checked for bleeding to ensure excellent blood supply. The repair is completed by suturing the rectal flap to the distal rectal wall using interrupted absorbable sutures.
At the end of the operation, the area is inspected for hemostasis. A dressing is placed over the perineum and secured with mesh panties. Rectal packing is not necessary. Most patients can be discharged same day as the operation with outpatient follow-up along with pain medication, stool softeners, and sitz baths.
Our patient had an uneventful postoperative course. The fistula was fully healed without any signs or symptoms of recurrence at 8 weeks follow-up exam. Her protective ileostomy was then successfully closed and she remains free of recurrence.
Results
Tips, tricks and pitfalls
As the rectal flap is developed, the width should gradually increase so that the base is at least twice the width of the apex to ensure adequate blood supply.
A clear plane in the rectovaginal septum should be visualized when dissecting the rectal flap free from the vagina. This can be achieved by first developing a plane proximal to the fistula opening and working laterally to medially on either side of the flap away from scar tissue. Those planes can then be connected at the apex of the flap, which contains the fistula opening, allowing for dissection in a bloodless plane and preserving the full vascular supply to the rectal flap.
The edges around the vaginal fistula opening should be debrided until healthy vaginal tissue is reached. If the surgeon chooses to close the vaginal opening, this should be done loosely with absorbable sutures to allow for some drainage from underneath the rectal flap in case a hematoma develops.
Conclusions
Transanal repair of a RVF through creation of a rectal advancement flap can be used to treat appropriately selected patients. We have demonstrated a surgical technique that utilizes vaginal wall plication to reinforce RAFR of a RVF in patients with significant tissue defects. Future studies are needed to further evaluate the efficacy of this technique.
Acknowledgements
The authors would like to acknowledge the James R. and Helen D. Russell Institute for Research and Innovation at Advocate Lutheran General Hospital.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study adhered to the prescribed ethical guidelines. Informed consent was obtained from the patient to use the video recording of her operation for educational purposes. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
References
- de Bernis L. Obstetric fistula: guiding principles for clinical management and programme development, a new WHO guideline. Int J Gynaecol Obstet 2007;99 Suppl 1:S117-21. [Crossref] [PubMed]
- Tsang CB, Madoff RD, Wong WD, et al. Anal sphincter integrity and function influences outcome in rectovaginal fistula repair. Dis Colon Rectum 1998;41:1141-6. [Crossref] [PubMed]
- Lowry AC, Thorson AG, Rothenberger DA, et al. Repair of simple rectovaginal fistulas. Influence of previous repairs. Dis Colon Rectum 1988;31:676-8. [Crossref] [PubMed]
- Debeche-Adams TH, Bohl JL. Rectovaginal fistulas. Clin Colon Rectal Surg 2010;23:99-103. [Crossref] [PubMed]
- de Parades V, Dahmani Z, Blanchard P, et al. Endorectal advancement flap with muscular plication: a modified technique for rectovaginal fistula repair. Colorectal Dis 2011;13:921-5. [Crossref] [PubMed]
- Hull TL, El-Gazzaz G, Gurland B, et al. Surgeons should not hesitate to perform episioproctotomy for rectovaginal fistula secondary to cryptoglandular or obstetrical origin. Dis Colon Rectum 2011;54:54-9. [Crossref] [PubMed]
- Jarrar A, Church J. Advancement flap repair: a good option for complex anorectal fistulas. Dis Colon Rectum 2011;54:1537-41. [Crossref] [PubMed]
- El-Gazzaz G, Hull TL, Mignanelli E, et al. Obstetric and cryptoglandular rectovaginal fistulas: long-term surgical outcome; quality of life; and sexual function. J Gastrointest Surg 2010;14:1758-63. [Crossref] [PubMed]
- Pinto RA, Peterson TV, Shawki S, et al. Are there predictors of outcome following rectovaginal fistula repair? Dis Colon Rectum 2010;53:1240-7. [Crossref] [PubMed]
- Ellis CN. Outcomes after repair of rectovaginal fistulas using bioprosthetics. Dis Colon Rectum 2008;51:1084-8. [Crossref] [PubMed]
- Uribe N, Millán M, Minguez M, et al. Clinical and manometric results of endorectal advancement flaps for complex anal fistula. Int J Colorectal Dis 2007;22:259-64. [Crossref] [PubMed]
- Mizrahi N, Wexner SD, Zmora O, et al. Endorectal advancement flap: are there predictors of failure? Dis Colon Rectum 2002;45:1616-21. [Crossref] [PubMed]
- Sonoda T, Hull T, Piedmonte MR, et al. Outcomes of primary repair of anorectal and rectovaginal fistulas using the endorectal advancement flap. Dis Colon Rectum 2002;45:1622-8. [Crossref] [PubMed]
- Zimmerman DD, Gosselink MP, Briel JW, et al. The outcome of transanal advancement flap repair of rectovaginal fistulas is not improved by an additional labial fat flap transposition. Tech Coloproctol 2002;6:37-42. [Crossref] [PubMed]
- Baig MK, Zhao RH, Yuen CH, et al. Simple rectovaginal fistulas. Int J Colorectal Dis 2000;15:323-7. [Crossref] [PubMed]
- MacRae HM, McLeod RS, Cohen Z, et al. Treatment of rectovaginal fistulas that has failed previous repair attempts. Dis Colon Rectum 1995;38:921-5. [Crossref] [PubMed]
- Sugrue J, Kaminski J, Marecik S, et al. Repair of a recurrent rectovaginal fistula using a rectal wall advancement flap reinforced with posterior vaginal wall plication under protection of a loop ileostomy. Asvide 2016;3:188. Available online: http://www.asvide.com/articles/944
Cite this article as: Sugrue J, Kaminski J, Patel S, Park J, Prasad L, Marecik S. Repair of a recurrent traumatic rectovaginal fistula using vaginal wall plication to reinforce a rectal wall advancement flap. J Vis Surg 2016;2:65.