Pain management using patient controlled anaesthesia in adults post Nuss procedure: an analysis with respect to patient satisfaction
Introduction
The option of surgical intervention for chest wall deformities, the commonest of which is pectus excavatum, is gaining popularity, as more novel minimally invasive surgical techniques are being developed. Initial treatment strategies for the correction of pectus excavatum relied upon more invasive and lengthy surgical procedures, the Ravitch procedure being the most common. The Ravitch procedure involves subperichondrial resection of the deformed costal cartilages and sternal osteotomy for fixation of the sternum anteriorly (1,2). In contrast, the Nuss procedure, a well-established minimally-invasive technique, requires only two small (approximately 2 cm) lateral incisions in either side of the chest wall. The procedure involves two interventions: the first, to insert the steel bar through the lateral thoracic incisions and the second, to remove the bar after 2 years. An endoscope is inserted into the chest cavity to guide the insertion of a metal bar, which is secured in its new position with stabilisers and wires. The patient must be anaesthetised prior to the operation. Increased severity of post-operative pain has been correlated with a greater Haller Index (a measure of the extent of the pectus deformity) (3). At our centre, we believe in the use of patient-controlled anaesthesia (PCA) as opposed to epidurals to decrease post-operative pain, hasten recovery and minimise complications and side-effects related to pain management.
Methods
Patient population
We carried out a retrospective survey on a total of 149 patients (139 males and 10 females), of average age 28, after undergoing the first intervention of the Nuss procedure- the initial concave bar insertion. The study was conducted between August and October, 2014. The survey assessed patient satisfaction with regards to pain management offered at hospital or sought after at home following hospital stay. The following questions were addressed in the survey: (I) Were you satisfied with the pain management you received in hospital immediately after your surgery? (II) Were you adequately warned about post-operative pain before the operation (i) by the surgeon? Or (ii) by the anaesthetist? (III) Did you have (i) more pain, (ii) less pain than expected? (IV) How effective was the medication prescribed for home usage at controlling the pain? (V) Did you need more medication? (VI) Did you need more pain-killers after the medicines we sent you home with had run out? (VII) Did you develop long-term or chronic pain? (VIII) How long did the pain last? Further data were also collated into a database of all pectus patients operated on at our institution to assist statistical analysis. This data includes the age of the patient, admission and discharge date, number and size of bar(s) inserted, location and number of stabilisers, medication on discharge, perioperative complications, postoperative pre-discharge complications, readmissions, reason for readmissions and pre-op and post-op images showing physical signs of correction.
Patient-controlled anaesthesia (PCA) technique
The outcome of our study was to measure patient satisfaction with postoperative analgesia and medication prescribed for home usage. Through this we aimed to prove the efficacy of PCA as a viable alternative to common intra-operative pain management strategies. Intra-operative pain management was administered through a multi-modal approach: morphine (10–20 mg total); intravenous paracetamol (1–2 g total); intravenous diclofenac (75 mg total) and bupivacaine (0.5% in wounds). The PCA technique post-operatively used a nurse-led protocol of a 1–2 mg bolus dose of morphine sulphate, delivered through a 5-minute lockout. There was no background infusion and no maximum dose limit in a 4-hour period. The pre-operative provision of information by the anaesthetist and surgeon, with regard to what to expect in terms of pain post-operatively, was also assessed.
Results
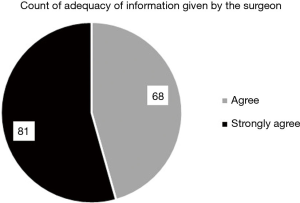
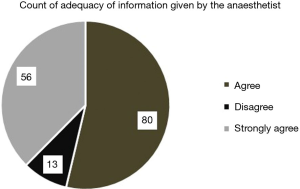
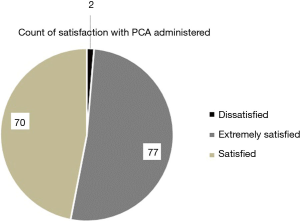
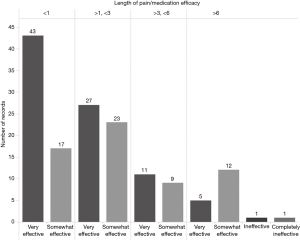
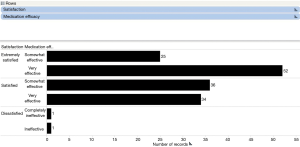
Centres who have shifted to an exclusively PCA regime exist in the minority. This study has proved that in a centre where all patients are managed exclusively using PCA patient satisfaction and medication efficacy is high, whereas there is a significant decrease in the length of pain. In Figure 1, the length of pain post-procedure, in months, is compared with patient assessment of immediate post-operative pain relief, on a 4-point scale, from very effective to completely ineffective. Patients who experienced less than 6 months of pain post-procedure all judged PCA to be either somewhat or very effective, with the majority judging it very effective. In those patients experiencing pain for less than 1 month post-procedure, more than twice as many judged PCA very effective versus somewhat effective. In the group of patients experiencing pain longer than 6 months, the majority of patients still judged PCA very or somewhat effective. Only two patients judged PCA ineffective out of the 19 in this group experiencing long-term pain. All patients agreed information given by the surgeon about postoperative pain managed by PCA was adequate, with the slight majority strongly agreeing rather than simply agreeing (Figure 2). Moreover, all but 2 patients out of the 149 questioned were satisfied with postoperative pain relief using PCA. Just over half the population [77] were extremely satisfied (Figure 3). Ninety-one percent of patients agreed that information given by the anaesthetist about postoperative pain controlled by PCA was adequate. This number is lower than the corresponding figure for the surgeon suggesting that it is important to have the anaesthetist present prior to procedure at the consultation stage (Figure 4). Finally, all patients were satisfied with the pain management received in hospital immediately after surgery and reported pain relief using PCA as being effective. More than twice as many who were extremely satisfied reported pain relief as being very effective than somewhat effective. For those who were simply satisfied with the procedure, numbers were similar in those reporting pain relief as somewhat or very effective. The two patients dissatisfied with the procedure were also the only two patients dissatisfied with the pain relief using PCA: one reporting it as completely ineffective and one simply ineffective (Figure 5).
There were no serious peri-operative complications or fatalities in any of the cases and all 149 procedures were successful.
Discussion
The minimally invasive Nuss procedure has become commonplace for the correction of pectus excavatum. The Nuss procedure is, however, associated with severe postoperative pain and the belief that thoracic epidural offers the best pain control has recently come under scrutiny after several studies have shown PCA to be just as effective. The patient population is predominantly young males with body-image issues associated with feelings of embarrassment, personal insecurity, depression and some perceptions of shortness of breath on exertion. Although there is evidence of restrictive lung function, the procedure remains mostly cosmetic and patient satisfaction is greatly correlated with post-operative aesthetic appearance. The cohort is generally highly-motivated and knowledgeable about the procedure as they have actively sought it. Therefore, the group is reasonably prepared for the degree of pain severity and have excogitated the procedure to the point where they are willing to go through with it regardless of possible long-term pain.
Inadequate pain management causes many post-operative complications, limits early mobilisation, limits enhanced-recovery and can prolong hospital stay. Contributing factors to the development of chronic pain include longer, more invasive interventions and severe patient-reported post-operative pain (4). Chronic pain has been reported in 67% of patients following thoracotomy, persisting in 25% of cases (5). A multi-institutional survey and review of pain management following the Nuss procedure revealed 91% of institutions to use thoracic epidural anaesthesia and the remaining 9% of surveyed institutions to use intravenous patient-controlled analgesia. However, 16% of the paediatric hospitals reviewed said that they were transitioning from using epidurals to opiate-based PCA techniques (6).
Intra-operative pain management options include thoracic epidural anaesthesia or an opiate based technique. Other methods that exist to manage pain post Nuss-procedure include lumbar spinal opiates, paravertebral nerve blocks, intercostal nerve blocks and hypnosis. The opiate technique involves a small dose of a short-acting opiate, such as fentanyl, combined with a longer-acting opiate, such as morphine, for a synergistic effect. Post-operative pain can be managed in recovery using incremental doses of intravenous fentanyl or morphine. During recovery, the nurses will monitor respiratory function and conduct sedation scoring until the patient reports no pain. PCA with fentanyl or morphine is a patient-led or nurse-led protocol, depending on the age of the patient. A standard PCA protocol includes a bolus dose (e.g., 1 mg of morphine or 20 μg of fentanyl) and a 5-minute lockout period, with or without a low dose background infusion, enabling the patient to titrate medication according to individual needs and perceptions of post-operative pain. There is a strong correlation between more severe pectus deformation and increased morphine use. PCA allows the patient greater control over their medication. Opiods are intravenously administered at a continuous rate allowing for the patient to gain easier access to the medication. Due to the lockout period there is also a decreased chance of medication error.
Another technique as previously mentioned are thoracic epidurals. Although thoracic epidurals are currently used in the majority of centres, many complications arise following their use. The major sequelae observed following the use of thoracic epidurals include, firstly inadequate analgesia and missed segments, and also intra-operative and post-operative hypotension alongside urinary retention and delayed mobilisation. Epidural haematomas and spinal cord ischaemia are frightening sequelae that demand fast neurological intervention that is not always present in centres offering the procedure (7,8). Recent UK data reports delayed diagnosis in 4 out of 5 cases of epidural haematomas. In addition, renal function must be closely monitored with patients receiving thoracic epidural anaesthesia. Catheter removal is also a critical phase in treatment lending to assiduous neurological monitoring to prevent epidural bleeding and decrease the chances of haematomas (9). The timing of the administration of thromboprophylaxis drugs differs amongst centres. Infusion of low-molecular weight heparin (LMWH) must be carefully timed in relation to the placement and removal of the epidural catheter. The infusion should not occur until 6 hours post catheter insertion and the catheter removal must take place when the levels of LMWH are at their lowest. These are all essential steps that must be taken by the thoracic anaesthetist to minimise haematoma complications and therefore if not followed correctly, there is a chance of error (10). The National Audi Project 3 (NAP3) carried out by the Royal College of Anaesthetists in the UK showed a complication rate of 4.2 in 100,000 central neuraxial blocks including spinals, epidurals and combine spinal-epidural techniques. Thirty permanent injuries were reported and 60% of these were in patients with epidurals. Complications occurred mostly with peri-operative epidurals (11).
Thoracic epidural anaesthesia requires laborious, intensive nursing monitoring on high-dependency units. It is, therefore, more costly than PCA. In addition, it is imperative that thoracic epidurals are placed when the patient is awake or only mildly sedated otherwise there is significant risk of neurological complications. In the Nuss procedure, this may not always be achievable as the patient population is relatively young.
In contrast, some studies have shown thoracic epidurals to be superior to PCA opiate analgesia in the immediate post-operative period (12). A randomised study comparing PCA to epidural use found epidural insertion to be wrongly placed in 22% of cases and for it to significantly prolong operative time (13). Due to the need for highly-experienced epidural lists, nursing staff and high-dependency units, it would be difficult to justify the use of thoracic epidurals in fit, young patients and we believe that PCA should become the gold standard amongst all institutions offering the Nuss procedure.
Conclusions
This study provides further incentive for centres to adopt PCA as the main pain management strategy in order to offer and achieve excellent pain control in the peri-operative period, achieve high rates of patient satisfaction and prevent instances of chronic pain. Further, we believe that the patient group would benefit from access to anaesthetists trained specifically in chronic pain management and a multi-disciplinary chronic pain service in the event of the development of post-operative chronic pain syndromes.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by the institutional ethical committee. Written informed consent was obtained from the patient for publication. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
References
- Nasr A, Fecteau A, Wales PW. Comparison of the Nuss and the Ravitch procedure for pectus excavatum repair: a meta-analysis. J Pediatr Surg 2010;45:880-6. [Crossref] [PubMed]
- Ravitch MM. The Operative Treatment of Pectus Excavatum. Ann Surg 1949;129:429-44. [Crossref] [PubMed]
- Castellani C, Schalamon J, Saxena AK, et al. Early complications of the Nuss procedure for pectus excavatum: a prospective study. Pediatr Surg Int 2008;24:659-66. [Crossref] [PubMed]
- Katz J, Jackson M, Kavanagh BP, et al. Acute pain after thoracic surgery predicts long-term post-thoracotomy pain. Clin J Pain 1996;12:50-5. [Crossref] [PubMed]
- Perttunen K, Tasmuth T, Kalso E. Chronic pain after thoracic surgery: a follow-up study. Acta Anaesthesiol Scand 1999;43:563-7. [Crossref] [PubMed]
- Muhly WT, Maxwell LG, Cravero JP. Pain management following the Nuss procedure: a survey of practice and review. Acta Anaesthesiol Scand 2014;58:1134-9. [Crossref] [PubMed]
- Scherer R, Schmutzler M, Giebler R, et al. Complications related to thoracic epidural analgesia: a prospective study in 1071 surgical patients. Acta Anaesthesiol Scand 1993;37:370-4. [Crossref] [PubMed]
- Rudin A, Flisberg P, Johansson J, et al. Thoracic epidural analgesia or intravenous morphine analgesia after thoracoabdominal esophagectomy: a prospective follow-up of 201 patients. J Cardiothorac Vasc Anesth 2005;19:350-7. [Crossref] [PubMed]
- Freise H, Van Aken HK. Risks and benefits of thoracic epidural anaesthesia. Br J Anaesth 2011;107:859-68. [Crossref] [PubMed]
- Horlocker TT. Regional anaesthesia in the patient receiving antithrombotic and antiplatelet therapy. Br J Anaesth 2011;107 Suppl 1:i96-106. [Crossref] [PubMed]
- Wildsmith JA. Continuous thoracic epidural block for surgery: gold standard or debased currency? Br J Anaesth 2012;109:9-12. [Crossref] [PubMed]
- Stroud AM, Tulanont DD, Coates TE, et al. Epidural analgesia versus intravenous patient-controlled analgesia following minimally invasive pectus excavatum repair: a systematic review and meta-analysis. J Pediatr Surg 2014;49:798-806. [Crossref] [PubMed]
- St Peter SD, Weesner KA, Weissend EE, et al. Epidural vs patient-controlled analgesia for postoperative pain after pectus excavatum repair: a prospective, randomized trial. J Pediatr Surg 2012;47:148-53. [Crossref] [PubMed]
Cite this article as: Kolvekar S, Pilegaard H, Ashley E, Simon N, Grant J. Pain management using patient controlled anaesthesia in adults post Nuss procedure: an analysis with respect to patient satisfaction. J Vis Surg 2016;2:37.