
Prospects of 3D-printed sternum prostheses: a review
Introduction
Large chest wall defects following resection of a chest wall tumor in ribs or sternum, have always been associated with higher morbidity and mortality. Both the better understanding of the physiology of breathing and the upcoming technical possibilities to integrate stable and inert materials in chest wall defects have led to drastic improvements in respiratory function, cosmetic results and thus the quality of life after chest wall resections.
Current concepts of reconstruction after full thickness chest wall resections are described as “sandwich technique” and include mesh-closure of the defect to prevent lung herniation, rigid stabilization to overcome flail chest and coverage with viable tissue, to secure sterile embedding of the foreign material and protective closure (1-3). Methyl-methacrylate has long been used with flexible meshes to create patient adapted rigid prostheses in situ with encouraging results (2). But the conjunction with rib stumps caused permanent pain or discomfort in some patients because a stable anchoring was not always possible.
Ideal materials to cover chest wall defects need to be remarkably versatile: rigid and stiff to protect the thoracic organs and prevent paradoxical motion on the one hand, malleable to adapt to the shape of the chest wall, flexible enough to allow breathing excursions, biologically inert or even allow the ingrowth of tissue and radiolucent on the other hand (4,5).
These requirements are only partially met with materials which are in widespread use at present (6-9).
The invention of 3D printing techniques with the possibility of producing exact titanium or polyethylene-based copies of virtually any part of the skeletal body has led yet to another era in reconstructive surgery (10,11).
The aim of this review is to gather experiences regarding these new materials with special focus on sternum resection and finally, to draw prudent conclusions as to if or how the treatment of rare chest wall tumors can be improved with new implants.
Indications
The necessity of a bone prosthesis for the chest wall obviously depends on the size as well as the location of the resection. While defects in the anterolateral region that are bigger than 5 cm in diameter or involving more than three ribs always need coverage, at least with artificial or biological mesh to avoid lung herniation, there is no need to cover even larger resections in the dorso-apical region because of the additional protection of scapula and shoulder girdle (7,12). Rigid reconstruction after sternal resection is mandatory at least for large resections >10 cm or complete sternectomy, because paradoxical respiration cannot be overcome with mesh implants alone (8).
Materials and methods
We entered the words—sternum prosthesis—in the PubMed database and compared all case reports in English language with the clear focus on new technologies, namely 3D printing in comparison to differently sized standard models. The process of 3D printing and characteristics of the particular material are then described. We found reports about customized 3D molds made from titanium, carbon fiber, and polyethylene with short- and mid-term results (5,10,11,13-19). Alumina ceramic was the chosen material for standardized sternum replacements (20,21). As a comparison, we included conventional sternum reconstruction with titanium bars into our analysis (1-3,6-9).
Titanium rib bridge system
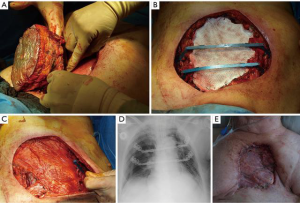
Fabre et al. presented a series of 24 patients with sternum resections of >10 cm who received chest wall reconstruction with a layer of mesh sutured to the rib ends and reinforcement of the chest with titanium rib bridge system (STRATOS, MedXpert, Germany) in 19 patients. They used a mean of 2 bars per patient. Similarly treated patients were presented in studies by Hamad et al., Berthet et al., De Palma et al., and Gao et al. (6,7,9,22). We present a case with similar reconstruction (Figure 1).
Titanium molds
Based on high resolution CT scan data, a 3D model of the bony thorax and tumor was generated using a special software. With an additional computer-assisted design (CAD) measurement tool, the 3D model of a prosthesis was designed. Another Program translated these data into a readable STL (Standard Triangle/Tessellation Language) format for the 3D printer. Using powdered titanium with the electron beam melting (EBM) technology, layer on layer of fused material was built into the complex titanium implant (10,13-19). Software and materials used are listed in the table below.
Each of the presented cases using customized sternum replacements planned a resection area of 1 to 3 cm to the edge of the tumor in order to achieve tumor-free margins. However, every model was developed individually to allow the best possible anchorage to the rib stumps while at the same time give space to intraoperative alterations on the margins.
Turna et al. developed a customized titanium sternum model in 2013 in a case of a resected superinfected metastasis of a breast carcinoma that extended to clavicles, ribs, and the complete sternal body. After the printing process, the model was sandblasted to ensure a better adherence to granulating tissue. Their prosthesis was comparatively flat with multiple burr holes on each arm and could easily be fixed and covered intraoperatively (14).
Demondion et al. presented their 3D-printed sternal titanium plate after a sternal resection due to bone metastasis. Their design was a simple customized plate with multiple perforations connected to three staples on each side, drawn to be tightened on the adjacent ribs (Figure 2) (13).
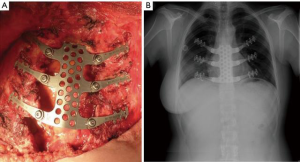
Aranda et al. designed their 3D implant as a rigid sternal core with titanium rods as neo-ribs which ended in attachment clamps. A second resection template was made for exact intraoperative margin planning. The titanium clamps were fixed with screws through the ribs and the core was then fixed with steel wire through existing holes (10).
Aragón et al. replaced the inferior half of the sternum with the adjacent three ribs with a 3D-printed customized titanium mold after resection of a chondrosarcoma in 2016. The costo-sternal joints were designed to allow dynamic movements similar to those in the natural articulation (15).
The 2017 sternum model of Moradiellos and colleagues was designed with a folding pattern allowing some expansion and contraction of the chest. This implant had foldable jaws that were clamped over the healthy ends of the resected ribs. The cranial and caudal ends were fixed to the sternal rest with screws (16).
Oswald et al. have altered the central portion of the titanium structure was coated in porous polyethylene to allow better colonization with granulation tissue. The arms of the prosthesis were shaped as two plates which were approximated with screws through the ribs. The sternal ends were fixed with steel wires. Since this patient initially had sternal osteomyelitis, the first repair after the sternal resection was made with a pedicled left pectoralis major flap. The definitive reconstruction with the 3D-printed prosthesis was performed 5 years later. Detailed lung-function-tests were run prior to and after the reconstruction (17).
In 2018 Ma et al. planned bigger holes on the sides of their sternal implant to pass through ligaments for fixation to the clavicles in order to preserve the mobility of the shoulder girdle. The ligaments were harvested from the patient’s own palmaris longus and semitendinosus muscles. For the residual basal part of the sternal corpus, inert lines were made onto the caudal bar of the model with additional burr holes for the bone cement to stabilize (18).
The sternum prosthesis of Wen et al. added a 2-cm fixing area to allow some intraoperative resection difference to the 3D plan, in order to secure fixation at the rib margins. The raw prosthesis was finally polished and sandblasted to create a rough granular surface. The last production step took three working days (19).
Carbon fiber molds
The custom-made carbon-fiber implant was used to reconstruct an 8 cm × 7 cm defect after extensive tumor resection of the sternum. A 3D electron model was constructed through CT data. The implant was then manufactured within 7 days using the carbon fiber 3D sintering printing technique. The implant was fixed to the rib stumps with cruciate ligament sutures and separately to the manubrium using steel wires. Carbon fibers have a low density of 1.5 g/cm3 and do not deflect X-rays and ionizing radiation. Furthermore, this implant has a microporous surface that facilitates the inward growth of adjacent tissue (5).
Alumina ceramic molds
The characteristics of alumina ceramic imply osteoconduction and osseointegration, biocompatibility, a certain level of bacterial resistance, radiotransparency, and mechanical strength. The material has been used for more than 20 years in orthopedic surgery. Alumina ceramic prostheses are available in 5 sizes with pre-drilled holes for non-resorbable suture threads to be anchored to the ribs. The ingrowth of the rib ends into the ceramic results in the stability of the chest. The implant sets are provided with differently sized metallic models to choose the best fitting ceramic implant after tumor resection. Six cases have been treated and reported so far (20). Loading of the prosthesis with antibiotics for use in infected wound surroundings was another case recently reported (21).
Polyethylene molds
The polyethylene manubrium implant was manufactured using numerically controlled milling machine, and was made of certified polyethylene of ultrahigh molecular mass. The design of the prosthesis was based on CT data. Polyethylene is rigid, non-absorbable, and yet the only material in use that is malleable after fabrication. Intraoperatively, the size of the implant was trimmed using a sharp knife and was fixed to the clavicles, sternum and ribs with the help of titanium rib plates (11).
Results
Short-term results
Sternum reconstruction is most often performed with titanium bars bridging the defect from one rib stump to the other. In the series of Fabre et al., a radical tumor resection (R0) was gained in all patients (8). All patients needed closure of the defect with polytetrafluorethylene (PTFE) patch or Vicryl to prevent lung herniation. A mean of two bars with rib clips were found to offer enough stability of the chest wall. There was no perioperative mortality. Wound infection did occur in one patient but did not require the removal of the titanium rib system. Postoperative FEV1 did not significantly differ from pre-operative values. The authors stated therefore that reconstruction with a titanium rib bridge system has low morbidity and permits a rapid return to baseline pulmonary mechanics. To compare different newer modalities of defect reconstruction, we categorized the results in Table 1.
Table 1
| Study | N | Age, years | Infected or non-infected sternum | Material of prosthesis | Covering | Software | Estimated costs | Production/preparation time | Follow-up | Complications |
|---|---|---|---|---|---|---|---|---|---|---|
| Hamad 2009 | 1 | 50 | Not infected: mamma Ca metastasis |
Titanium bars, Gore tex mesh | MPM | None | Not stated | Ready-to-use | 14 months | None |
| Berthet 2011 | 19 | 34–72 | Not infected: all neoplastic; 11 NSCLC; 5 sarcoma; 1 breast Ca; 1 thyroid Ca; 1 basal cell Ca |
Titanium plates and dual mesh | MPM or LDM 9; omentum 2 | None | Not stated | Ready-to-use | 30 months | 1 death of PE; 2 deep infections; 1 partial skin necrosis; 2 delayed ruptures of titanium bars |
| Fabre 2012 | 24 | 31–85 | Not infected | Vicryl mesh 14; PTFE mesh 5; Stratos bars all | MPM 12; LDM 9 | None | Not stated | Ready-to-use | 26 months | Reintubation 1; tracheostomy 1; lung infection 1; ARDS 1, seroma 2 |
| Demondion 2014 | 1 | 28 | Not infected: breast cancer metastasis | Custom-made 3D-printed titanium prosthesis | MPM | Thorib®, NeuroFrance Implants | Not stated | Not stated | 6 months | None |
| Turna 2014 | 1 | 62 | Infected: breast cancer metastasis | Custom-made 3D-printed titanium prosthesis | LDM + split thickness graft | 3D: Mimics 16.0 Digital design: 3-matic 8.0, Magic RP 17.02, MEDMC (Medical Design and Manufacturing Center, GATA). SLM-machine (M2 cusing, Concept Laser GmbH, Lichtenfelds, Germany) | Not stated | Not stated | 3 months | None |
| Aranda 2015 | 1 | 54 | Not infected: chondrosarcoma | Custom-made 3D-printed titanium prosthesis | Dual mesh to the rear side, myocutaneous LDM | Anatomics Pt Ltd. (St Kilda, Australia) | Not stated | 1 month | 12 days | None |
| Hummelink 2015 | 1 | 59 | Not infected: fibrous dysplasia | A patch made of two polypropylene meshes and methyl methacrylate | Subcutaneous tissue + skin | None | Not stated | Ready-to-use | 6 weeks | None |
| Aragón 2016 | 1 | 57 | Not infected: chondrosarcoma | Cust-made 3D-printed titanium prosthesis with polypropylene mesh | Externally: not stated | InVesalius (ACV. External SL. Madrid); Arcam Q10 | Not stated | Not stated | 3 months | None |
| De Palma 2016 |
27 | 16–83 | Not infected: 11 neoplastic; 15 traumatic; 1 malformation |
Synthes® titanium plates system; 8 patients: + Mersilene mesh | All: MPM, LDM, anterior serratus, abdominis obliquus ext. | None | Not stated | Ready- to-use |
1–59 months | 15 none; 10 minor complications; 2 major complications: pneumonia 1; sepsis with death 1. Late c. plate ruptures 2, pin dislodgement 1 |
| Lipińska 2017 |
1 | 52 | Not infected: renal cancer metastasis | Custom-made polyethylene prosthesis | MPM | SolidWorks program, solid form with “parasolisd.xt” extension was achieved in “NX CAM” generating special codes for numerically controlled milling machine |
Not stated | 7 days | 6 months | None |
| Moradiellos 2017 | 1 | 32 | Not infected: NUT carcinoma | Custom-made 3D-printed titanium prosthesis | Dualmesh internally; MPM externally | Not stated | Stated as “remaining concerns for this technique” | Stated as “remaining concerns for this technique” | 6 months | None |
| Sheikhy et al. 2017 | 1 | 20 | Sterile; aneurysmal bone cyst | Cryopreserved sternal allograft | Breast tissue, no muscle | None | Not stated | 1 month | 18 months | None |
| Marulli 2017 | 18 | 35–78 | Sterile: 16 neoplastic; 2 sternotomy dehiscence | Cryopreserved sternal allograft | MPM 11; MPM + LDM 2; LDM 1 | Nonefig | Not stated | – | 11–84 months | R1, R2 resection 1 each; 1 death of PE; 1 fungal infection; 1 bleeding muscle flap; 1 screw removal |
| Oswald 2018 | 1 | 54 | Resected osteomyelitis with muscle flap | Custom-made 3D-printed titanium prosthesis with central polyethylene coating | MPM | Not stated | Not stated | Not stated | 14 months | None |
| Ma 2018 | 1 | 56 | Not infected; chondrosarcoma | Custom-made 3D-printed titanium prosthesis | MPM | Mimics 19.0 converted to STL format and imported into a laser metal direct-forming 3D printing device | Not stated | Not stated | 3 months | None |
| Bertin 2018 | 6 | 55–79 | 5 sterile; 1 infected (DSWI) | Porous alumina ceramic | None stated | None | Not stated | Ready-to-use | 20 months (3–37 months) | 1 Hematoma 2 superficial wound infections |
| Wen 2018 | 2 | 62 and 64 | Not infected | Customized titanium 3D printed rib and sternum prostheses |
MPM; M. rectus abdominis flap | Mimics software Digisurf (3D Family Corporation, Taiwan). The 3D model: ZPrinter® 310 Plus, (Z Corporation, Cambridge, MA, USA) | $1,200 for two ribs and $1,300 for the sternum | – | 6 months | None |
| Gao 2018 | 6 | 38–64 | Not infected all malignant | Titanium bars + polyester fabric | Not stated | None | Not stated | Ready-to-use | 10 months | 1 superficial wound infection |
| Wang 2019 | 1 | 61 | Not infected: synovial sarcoma | Custom-made carbon fiber | Not stated | Mimics software; Carbon fiber 3D sintering printing technique (Tankang biotech, Changsha, China) | Not stated | 7 days | 24 months | None |
| Tricard 2019 | 1 | 68 | Infected osteomyelitis | Gentamycin-loaded porous alumina | None stated | None | Not stated | Ready-to-use | 24 months | None |
Ca, carcinoma; MPM, major pectoralis muscle; NSCLC, non-small cell lung cancer; LDM, latissimus dorsi muscle; PE, pulmonary embolism; PTFE, polytetrafluorethylene; ARDS, acute respiratory distress syndrome; ext., externus; STL, Standard Triangle/Tessellation Language; late c., late complication; DSWI, deep sternal wound infection; SLM-machine, selective metal laser sintering machine; Mimics, image processing software for 3D design and modeling (Materialise®, Leuven, Belgium).
Standard reconstructions were reported in higher numbers and were associated with perioperative complications and mortality. Pulmonary embolism, acute respiratory distress syndrome (ARDS), superficial and deep wound infections as well as breakdown of the titanium reconstructions were described in around 0–20% of the cases (7-9).
The six patients studied by Gao et al. were operated using a Synthes Titanium fixation system and mesh and were reported to have returned to their daily (working-) life between 2 to 8 weeks after surgery. Their level of satisfaction with the functional and cosmetic results were stated very high with the exception of one patient who ranked his satisfaction with 6 out of 10 points (22).
All reported cases of sternal resections due to underlying malignancy, reported tumor-free resection margins. Apart from a localized seroma in the bed of the latissimus dorsi muscle and prolonged superficial wound healing, no major complications were reported with either of the above-mentioned 3D-printed implants. The patients recovered quickly after the operation and were reported to have little to no chronic pain or loss of lung function. As there are only individual case reports, statistical statements are not possible.
Long-term results
Since nearly all of the presented reports were on oncological chest wall resections, recurrent disease or metastatic spread in other bone structures was often reported before a long-term follow-up could be achieved. Especially after the resection of bone metastases, the recurrence was in some cases less than 4 months (6,10,11,13-16,18).
A 3D-printed carbon-fiber prosthesis after a sarcoma resection in a 61-year-old by Wang et al. was reported to have been stable for 24 months (5). A 14-month follow-up was achieved by Oswald et al. with their patient suffering from osteomyelitis, who received the polyethylene-coated sternum prosthesis. Because this man had previously been aided with a pectoralis flap, detailed function tests were run to compare the range of exercises possible with a soft tissue reconstruction of a sternal defect to before and after a solid sternal replacement. The differences in lung capacity, breathing frequency and FEV1 were significantly better after the rigid metal replacement (17).
The longest follow-up period with alumina ceramic sternum implant was 37 months without major complication. The CT scans revealed an ingrowth of the adjacent bone structures into the porous material after 4 months (20).
The larger patient cohorts of Berthet, De Palma and Fabre were all treated in the standard technique for their chest wall defects (7-9). They presented higher complication rates, however, their patients suffered from partially advanced malignancies, severe co-morbidities and/or older age. Interestingly, there was a delayed midline-rupture of two titanium plates used after traumatic sternum fracture (9) and two fractured titanium bars in Berthet’s series (7). They used the Stratos system to stabilize the chest wall defects. Two of the patients suffered from a delayed rupture of the titanium bar at the joint. In one patient, the bar was removed 6 months after the original surgery without replacement. The other patient had a ruptured and superinfected plate that needed removal on day 10 and intensive wound cleansing. One other case of superinfected osteosynthesis material was reported in this study that needed removal. One patient of De Palma’s cohort had to be re-operated on to remove the plate. The other patients fractured plate was solely a radiological finding without any clinical complaints that needed no re-intervention until the last follow-up at 35 months after the surgery.
In the authors experience, the rupture of a Stracos clamp has been seen in one obese patient with chronic obstructive pulmonary disease (COPD) under steroid treatment, one dislocated sternal plate was seen in another patient with severe kyphosis due to osteoporosis.
Costs
Unfortunately, the expenses for the proposed techniques have not been stated in all but one case report (19). The costs of the 3D-printed models obviously depend on size and thickness of the prostheses, so does the time required to produce them. Since it is mostly the 3D-printing process itself that amounts for most of the costs, the material plays a less significant role in the monetary calculations. The price for one alumina prosthesis that comes in ready-to-use kits costs 10,000–15,000 € to the best of the authors’ knowledge. Most cost-efficient is the standard treatment method with a combination of titanium bars and mesh; each titanium clip and bar (Stracos MedXpert) costs between 400 and 500 € to the authors experience.
Discussion
The aim of any oncological chest wall resection should be the complete removal of the tumor with free resection margins followed by a stable reconstruction that allows an uncomplicated wound healing with minimal functional impairment and good cosmetic results while observing cost efficiency. Both the conventional as well as the newer techniques discussed here fulfilled these aims (1-25).
With the emergence of titanium bars and availability of numerous organic tissues as well as inorganic substitutes, thoracic surgeons were given the tools to reconstruct most chest wall defects. Numerous studies proved a variety of these materials to be feasible even for larger resection holes on the chest wall with involvement of the sternum (6-9). The intraoperatively reformable titanium bars and plates provide a cost-effective, flexible, ready-to-use solution with a short learning curve (Video 1, chest wall resection and reconstruction with mesh, titanium plate and muscle flap). Disadvantages are the smooth surface and the lack of stability as was shown in the reports of Berthet and De Palma with ruptured plates and bars. The fractured plates mentioned earlier demonstrate dramatically the great powers affecting the sternum or its replacement in daily life. Lung function tests of other reported cases prove the positive outcome of a robust stabilization of the rib cage (7,9).
However, it is important to note that the patients in Berthet’s study in 2011 were by far the most crucial with severe co-morbidities, such as neoadjuvant radio-chemotherapy, advanced malignancies and old age.
With the necessity of a mesh graft to cover the thoracic defect, the risk of seroma with subsequent infection seems to be higher compared to the 3D-printed models that did not require further coverage of the thoracic organs. In addition, lacking bone substance due to osteoporosis impairs a stable fixation of the standard fixation systems.
Sternal allografts
Compatible allografts of deceased donors have been successfully transplanted to cover large chest wall defects. Months after the operation, the osseous tissue of the recipient patients overgrew on the transplant and resulted in a stable chest wall. The studies and case reports available prove the feasibility of this method (23-25). However, the availability and storage in tissue banks as well as the time-consuming processing until the graft is usable are surely less convincing, not to mention the paucity of organ donors.
Titanium molds
The advantages of titanium are its inert features, biocompatibility, flexibility, malleability and stability. The disadvantages are the price of the printing and its smooth surface, which does not allow surrounding tissue to adhere sufficiently if not sandblasted. One clear shortcoming of titanium is its impermeability for X-rays and the disruption of radiation therapy. Due to the cumbersome printing technique, the production can be very time consuming; the bigger the requested pieces are, the longer it would take the machine to form one layer over the other, thereby increasing the cost.
Leaving the monetary issue aside, the time needed to produce a larger mold is far from being acceptable in cases of oncologic indications: Aranda’s titanium sternum prosthesis took about 1-month manufacturing time (10).
Interestingly, Wen et al. reported relatively fair costs for customized rib and sternal body implants made of titanium compared to the current systems being used (19). Obviously, the materials, time and costs needed for a replica depend on its height and total size. Their customized sternal body and rib prostheses were relatively flat and hence only took 3 working days to produce. Wang et al. described a sternal prosthesis made of carbon-fiber that took seven days to be manufactured (5).
A porous polyethylene coating in the central part of the manubrium portion of the sternum prosthesis of Oswald et al. might enable the surrounding tissue to grow in and become one stable layer. Their model was implanted in a patient suffering from osteomyelitis of the manubrium-sternal junction and showed good mid-term results (17).
Carbon-fiber molds
Carbon-fiber does not disrupt X-rays or radiation beams due to its lower density. Its microporous structure allows surrounding tissue to grow in, making the graft more stable (5). The production process is similar to that of the titanium molds, hence the higher costs and long production period remain as clear limitation.
Polyethylene
Unlike all other materials mentioned above, polyethylene can be trimmed during the operation with a sharp knife and a surgical file, facilitating minor adjustments according to the specific requirements of the patient. Even though the material should be less costly than others mentioned here, the expensive 3D-printing process remains a shortcoming.
Aluminum ceramic mold
The ready-to-use sternum replacements proved to be suitable solutions for sternum reconstruction. The porous structure allows the ingrowth of tissue. If loaded with gentamycin, it shows promising use even in infected environment. Nevertheless, the costs are quite high and resection areas that involve parts of the ribs cannot be covered (20,21).
Bertin et al. proved their ready-to-use set of differently sized sternum replacements to be a suitable solution after sternum resections. The alumina ceramic material allows the surrounding tissues to grow in and form a solid bony mass, which was illustrated by impressive CT scan images 3 months after the implantation (20). Even in an infectious environment after osteomyelitis of the sternum, the gentamycin-coated version of the model is promising (21). Nevertheless, the costs, compared to standard techniques, are quite high, and resection areas involving parts of the ribs cannot be covered.
Even though the existing case reports on 3D-printed prostheses for large chest wall defects are rather scarce, the results are promising. With the 3D-image reconstruction of a chest CT, the resection margins can be precisely defined to shape a perfectly fitting mold that fills the surgical gap, thus preserving the structure and function of the thorax. Furthermore, the post-operative infection rates are low and the recovery of the cases that were treated with a personalized sternum prosthesis presented here was remarkably fast. Moreover, all patients could be discharged within 12 days and there was hardly any impairment in terms of lung function or mobility despite the vast resection of the rib cage (5,10,11,13-21).
Conclusions
The new 3D-printing technique offers endless possibilities in many fields, such as sternum replacements, solving surgical problems, and must be explored further. With the single cases reported on sternal prosthesis, one of the earliest successful use of this technique has been proven. However, the given examples are far from being the perfect solution. Many questions still remain, such as which design should be used that is more intraoperatively modifiable to achieve a stable yet flexible result, which material best meets the often-contradictive requirements.
The technique promises reductions in cost and production time with more routine and advances in technological development. Surgeons should play a vital part in giving input to ensure their interests are being considered in further developments.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jovs.amegroups.com/article/view/10.21037/jovs.2019.10.05/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the Helsinki Declaration (as revised in 2013). The manuscript is waived from patient informed consent according to the ethics committee or institutional review board.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Hummelink K, Ijsseldijk MA, Bosch AM, et al. Resection of a bony tumour of the chest wall with direct reconstruction using a sandwich technique: a standard technique for closure of large chest wall defects. J Surg Case Rep 2015; [Crossref] [PubMed]
- Mansour KA, Thourani VH, Losken A, et al. Chest wall resections and reconstruction: a 25-year experience. Ann Thorac Surg 2002;73:1720-5; discussion 1725-6.
- Chiappetta M, Facciolo F. Sternum reconstruction using titanium plates matched with "sandwich" Gore-Tex meshes. J Vis Surg 2018;4:47. [Crossref] [PubMed]
- Seder CW, Rocco G. Chest wall reconstruction after extended resection. J Thorac Dis 2016;8:S863-71. [Crossref] [PubMed]
- Wang B, Guo Y, Tang J, et al. Three-dimensional custom-made carbon-fiber prosthesis for sternal reconstruction after sarcoma resection. Thorac Cancer 2019;10:1500-2. [PubMed]
- Hamad AM, Marulli G, Bulf R, et al. Titanium plates support for chest wall reconstruction with Gore-Tex dual mesh after sternochondral resection. Eur J Cardiothorac Surg 2009;36:779-80. [Crossref] [PubMed]
- Berthet JP, Canaud L, D'Annoville T, et al. Titanium plates and Dualmesh: a modern combination for reconstructing very large chest wall defects. Ann Thorac Surg 2011;91:1709-16. [Crossref] [PubMed]
- Fabre D, El Batti S, Singhal S, et al. A paradigm shift for sternal reconstruction using a novel titanium rib bridge system following oncological resections. Eur J Cardiothorac Surg 2012;42:965-70. [Crossref] [PubMed]
- De Palma A, Sollitto F, Loizzi D, et al. Chest wall stabilization and reconstruction: short and long-term results 5 years after the introduction of a new titanium plates system. J Thorac Dis 2016;8:490-8. [Crossref] [PubMed]
- Aranda JL, Jiménez MF, Rodríguez M, et al. Tridimensional titanium-printed custom-made prosthesis for sternocostal reconstruction. Eur J Cardiothorac Surg 2015;48:e92-4. [Crossref] [PubMed]
- Lipińska J, Kutwin L, Wawrzycki M, et al. Chest reconstruction using a custom-designed polyethylene 3D implant after resection of the sternal manubrium. Onco Targets Ther 2017;10:4099-103. [Crossref] [PubMed]
- Kuzdzal J. ESTS Textbook of Thoracic Surgery. Chapter 62: Chest wall. Cracow: Medyzyna Praktyczna, 2014.
- Demondion P, Mercier O, Kolb F, et al. Sternal replacement with a custom-made titanium plate after resection of a solitary breast cancer metastasis. Interact Cardiovasc Thorac Surg 2014;18:145-7. [Crossref] [PubMed]
- Turna A, Kavakli K, Sapmaz E, et al. Reconstruction with a patient-specific titanium implant after a wide anterior chest wall resection. Interact Cardiovasc Thorac Surg 2014;18:234-6. [Crossref] [PubMed]
- Aragón J, Pérez Méndez I. Dynamic 3D printed titanium copy prosthesis: a novel design for large chest wall resection and reconstruction. J Thorac Dis 2016;8:E385-9. [Crossref] [PubMed]
- Moradiellos J, Amor S, Córdoba M, et al. Functional Chest Wall Reconstruction With a Biomechanical Three-Dimensionally Printed Implant. Ann Thorac Surg 2017;103:e389-91. [Crossref] [PubMed]
- Oswald N, Senanayake E, Naidu B, et al. Chest Wall Mechanics In Vivo With a New Custom-Made Three-Dimensional-Printed Sternal Prosthesis. Ann Thorac Surg 2018;105:1272-6. [Crossref] [PubMed]
- Ma XL, Wang DB, Ma JX, et al. Custom-made Prosthesis for Reconstruction after Radical Resection for Chondrosarcoma of Manubrium. Orthop Surg 2018;10:272-5. [Crossref] [PubMed]
- Wen X, Gao S, Feng J, et al. Chest-wall reconstruction with a customized titanium-alloy prosthesis fabricated by 3D printing and rapid prototyping. J Cardiothorac Surg 2018;13:4. [Crossref] [PubMed]
- Bertin F, Piccardo A, Denes E, et al. Porous alumina ceramic sternum: A reliable option for sternal replacement. Ann Thorac Med 2018;13:226-9. [Crossref] [PubMed]
- Tricard J, Chermat A, Denes E, et al. Antibiotic-loaded ceramic sternum for sternal replacement in a patient with deep sternal wound infection. Interact Cardiovasc Thorac Surg 2019; [Epub ahead of print]. [Crossref] [PubMed]
- Gao E, Li Y, Zhao T, et al. Reconstruction of anterior chest wall: a clinical analysis. J Cardiothorac Surg 2018;13:124. [Crossref] [PubMed]
- Dell'Amore A, Cassanelli N, Dolci G, et al. An alternative technique for anterior chest wall reconstruction: the sternal allograft transplantation. Interact Cardiovasc Thorac Surg 2012;15:944-7. [Crossref] [PubMed]
- Marulli G, Dell'amore A, Calabrese F, et al. Safety and Effectiveness of Cadaveric Allograft Sternochondral Replacement After Sternectomy: A New Tool for the Reconstruction of Anterior Chest Wall. Ann Thorac Surg 2017;103:898-905. [Crossref] [PubMed]
- Sheikhy K, Abbasi Dezfouli A, Sadegh Beigee F. Reconstruction of Chest Wall by Cryopreserved Sternal Allograft after Resection of Aneurysmal Bone Cyst of Sternum. Case Rep Surg 2017;2017:9135657. [Crossref] [PubMed]
- Balke D, Gupta V, Welter S. Open en bloc chest wall resection including three ribs and the right lower lobe for T3 lung cancer. Asvide 2020;7:024. Available online: http://www.asvide.com/watch/33041
Cite this article as: Balke D, Gupta V, Welter S. Prospects of 3D-printed sternum prostheses: a review. J Vis Surg 2020;6:10.


