Corporal excavation and penile prosthesis placement for severe corporal fibrosis
Introduction
The penile prosthesis is a standard treatment for men with erectile dysfunction refractory to vacuum, medical, or injection therapies. In patients with virgin tissue planes and normal anatomy, prosthesis placement can be technically straight forward and associated with minimal morbidity. Patients with corporal fibrosis, however, can present a unique surgical challenge for implantation. The process of corporal fibrosis starts with an initial insult to the corporal tissue. This triggers an inflammatory response that causes myofibroblast proliferation, collagenization, and eventual loss of functional cells. The replacement of normal corporal smooth muscle with this fibrotic, inelastic tissue prevents corporal expansion necessary to obtain an erection. On physical exam, this molecular process manifests as corpora with a hard, woody feeling. The initial insult causing corporal fibrosis has been associated with a variety of both acute and chronic conditions including aging, diabetes, heavy smoking, removal of an infected penile implant cylinder, priapism, penile trauma, Peyronie’s disease, or previous use of intracavernosal injection therapy (1-5).
Prosthesis implantation in patients with corporal fibrosis is one of the most difficult procedures in prosthetic urology and is associated with a higher risk of implant failure and infection compared to primary implantation (6,7). The most technically challenging component of this procedure is creating space within the corpora to allow appropriate accommodation and expansion of the implant cylinder. To facilitate implantation, many surgical approaches have been suggested such as corporal excavation, resection of scar tissue, use of cavernotomes, performing extended corporotomies, and the use of grafts for cylinder coverage (8,9). There is no standardized approach to prosthesis implantation in this setting, therefore selection of the appropriate surgical technique can vary depending on the extent of corporal fibrosis and surgeon experience.
Patient selection and workup
Our patient is a 39-year-old male with a history of hypertension, Type 2 insulin dependent diabetes mellitus, liver cancer, and erectile dysfunction that was referred to our clinic for penile prosthesis placement. His history included two previous episodes of prolonged priapism, each time managed with a distal shunt. Both priapism episodes were thought to be related to an adverse effect of trazodone. Prosthesis placement was attempted by his local urologist but was aborted intraoperatively due to severe corporal fibrosis. Following surgery, his post-operative course was complicated by a wound infection at the penoscrotal incision that was successfully treated with antibiotics and local wound care. He was subsequently referred to our reconstructive urology practice to explore further treatment options. At the time of our initial evaluation, the patient endorsed complete erectile dysfunction with an IIEF-5 of 1 out of 25.
The patient’s family history is notable for diabetes in both parents. He is single and a current cigarette smoker with 12.5 pack/year history. On physical exam his penis revealed woody fibrosis with hardened, atretic, inelastic corporal bodies bilaterally along their entire course. Due to the patient’s severe and extensive corporal fibrosis, he was offered a staged procedure in which we would attempt placement of a malleable prosthesis, followed by a three piece inflatable penile prosthesis (IPP) 3–6 months later. This treatment plan was decided upon due to his significantly increased risk of infection as well as high degree of technical challenge (7,10).
Given the severity of the patient’s fibrosis and the need to expose the corpora along its entire length, we opted for a perineal incision with invagination of the penis through the perineal incision. This approach was derived from our experience with complex urethroplasty. The patient was extensively counseled that this would be a difficult procedure that had a high risk of infection. The patient acknowledged all benefits and risks of the procedure and chose to proceed with the treatment plan as outlined above.
Pre-operative preparation
Our standard pre-operative work-up for patients undergoing penile prosthesis surgery includes CBC, BMP, urinalysis, urine culture, and HbA1c. Our patient’s pre-operative work-up was normal and his pre-operative HbA1c was 7%. Prior to surgery, weight-based intravenous vancomycin and gentamycin were administered within 1 hour of initial incision. Following induction of general anesthesia, the patient was placed in a simple lithotomy position. The skin of the suprapubic region, scrotum, and perineum was clipped and prepped with chlorhexidine scrub followed by a chlorhexidine and alcohol (Chloraprep) paint.
Equipment preference card
- Penile Prosthesis tray;
- Suture 3-0 Silk pops for stay suture, 2-0 PDS for corporotomy closure, 3-0 Vicryl and 4-0 Monocryl;
- Jordan Perineal Bookwalter;
- Tachosil Collagen Fleece;
- 9.5 mm Coloplast Genesis malleable implant (very important);
- Bacitracin antibiotic irrigation;
- Antimicrobial solution consisting of 1 g vancomycin, 3.375 g piperacillin/tazobactam, and 50 mg amphotericin B to soak the implant on the back table prior to insertion;
- 16 Fr Foley catheter.
Procedure
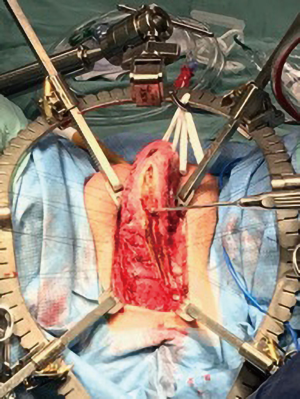
Our standard practice is to create an artificial erection by injecting 50 cc of sterile saline and 10 cc of 2% lidocaine as it both helps dilate the corpora and provides excellent local anesthesia. Due to the extreme nature of this patient’s corporal fibrosis, we were unable to inject the solution. A midline perineal incision was made and we carried out our dissection of the corpora and urethra en bloc. The penis was then invaginated into the perineal wound and placed on traction with a Penrose drain, exposing both corpora along their entire length (Figure 1).
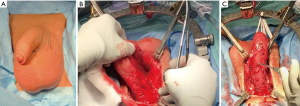
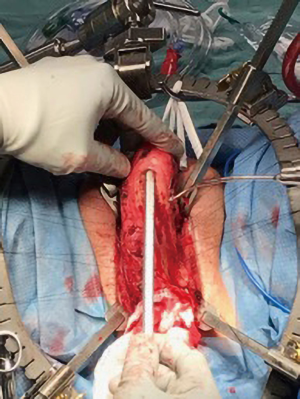
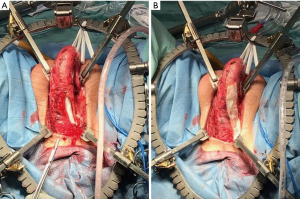
3-0 silk sutures were used to mark the medial and lateral edges of the left corporal body. Then Bovie cautery was used to make a vertical corporotomy along the full length of the left corpora (Figure 2). There was no bleeding from within the corporal tissue. The lumen was too narrow to accept even the smallest diameter penile implant, indicating our predicted need for extensive corporal excavation. We performed this by using a combination of both sharp and Bovie electrocautery dissection, taking extra care to remove the corporal scar from the inner surface of the tunica albuginea (Figure 3).
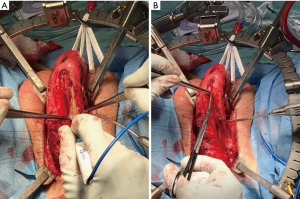
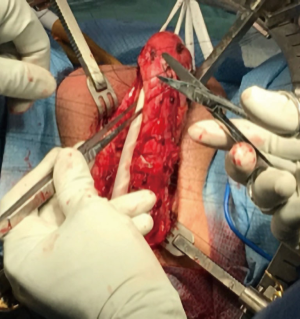
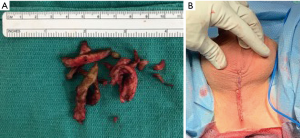
Once the diseased and fibrotic corporal tissue was removed, we then turned our attention to implant placement. A Furlow device was used to measure a corporal length of 14 cm (Figure 4). We elected to use a 13 cm, 9.5 mm Genesis malleable prosthesis. We closed the corporotomies with 2-0 PDS suture in a running fashion. For reinforcement of the closure, we overlaid Tachosil, a self-adhesive collagen fleece (Figure 5). We then repeated the same procedure on the right side (Figure 6).
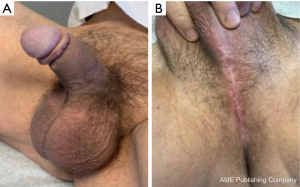
Following completion of the right side, the penis was reduced to its anatomic position. The cosmetic appearance was excellent and both implant tips were symmetrical and palpable in the mid glans. The incised fibers of the bulbocavernosus muscle were then reapproximated to the corpora bilaterally. The perineal wound was copiously irrigated with antibiotic solution and closed in standard fashion (Figure 7). The patient was awoken from anesthesia and transferred to the recovery room in stable condition.
Role of team members
- Aaron C. Lentz, MD—Attending surgeon;
- Steven Brousell, MD—Resident surgeon;
- Jason C. Chandrapal, MD—Videographer/Photographer and Urology Resident;
- Gregory J. Barton, MD—Videographer/Photographer and Urology Resident.
Post-operative management
The patient was admitted to the urology service and had an uncomplicated postoperative course. His Foley catheter was removed on postoperative day 1 and he was discharged home comfortably on a multimodal oral pain regimen and five days of broad-spectrum antibiotic prophylaxis, which is our standard practice.
On postoperative follow-up, approximately 7 weeks following his procedure, the patient had no complaints and stated that he was able to ejaculate without difficulty. His perineal incision was well healed with no signs of infection or wound dehiscence. The corporal cylinders were in place with symmetrical implant tips that were palpable in the mid glans and no sign of cylinder cross-over or impending erosion (Figure 8). Given his excellent postoperative outcome, the decision was made to continue with the previously planned second stage of the procedure, removal and replacement with a three-piece IPP, in 5–6 months. The entire operative video and follow-up can be viewed in Figure 9.

Tips, tricks, and pitfalls
- Careful pre-operative treatment planning guided our decision to utilize a malleable prosthesis. This was driven by the degree of corporal fibrosis and the concern for a high risk of complication if three-piece IPP placement was attempted.
- Utilization of the technique of penile invagination allowed us to achieve complete longitudinal corporal exposure while avoiding multiple incisions or overlapping incision near the corporotomy and cylinders.
- Tachosil or other biologic graft should be available for use in cases where the surgeon has suspicion of a difficult primary corporotomy closure.
- A staged approach to placement of a three-piece implant 3–6 months following malleable placement may maximize corporal length and reduce unwanted complications.
Conclusions
Penile prosthesis surgery in patients with severe corporal fibrosis can be one of the most challenging procedures in prosthetic urology. While many various surgical techniques have been previously described, there is no current standard surgical approach. We demonstrated that perineal penile invagination, corporal excavation, and implantation with a malleable prosthesis is a safe and effective treatment for these patients.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Martin Gross, Jay Simhan and Faysal A. Yafi) for the series “Penile Prosthesis Surgery” published in Journal of Visualized Surgery. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jovs.2019.07.03). The series “Penile Prosthesis Surgery” was commissioned by the editorial office without any funding or sponsorship. ACL reports other from Coloplast Corporation (speaker and consultant), other from Boston Scientific (speaker and consultant), outside the submitted work. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Martinez-Salamanca JI, Mueller A, Moncada I, et al. Penile prosthesis surgery in patients with corporal fibrosis: a state of the art review. J Sex Med 2011;8:1880-9. [Crossref] [PubMed]
- Gonzalez-Cadavid NF. Mechanisms of penile fibrosis. J Sex Med 2009;6:353-62. [Crossref] [PubMed]
- Ferrini MG, Kovanecz I, Sanchez S, et al. Long-term continuous treatment with sildenafil ameliorates aging-related erectile dysfunction and the underlying corporal fibrosis in the rat. Biol Reprod 2007;76:915-23. [Crossref] [PubMed]
- Kovanecz I, Ferrini MG, Vernet D, et al. Pioglitazone prevents corporal veno-occlusive dysfunction in a rat model of type 2 diabetes mellitus. BJU Int 2006;98:116-24. [Crossref] [PubMed]
- Tostes RC, Carneiro FS, Lee AJ, et al. Cigarette smoking and erectile dysfunction: focus on NO bioavailability and ROS generation. J Sex Med 2008;5:1284-95. [Crossref] [PubMed]
- Henry GD, Donatucci CF, Conners W, et al. An outcomes analysis of over 200 revision surgeries for penile prosthesis implantation: a multicenter study. J Sex Med 2012;9:309-15. [Crossref] [PubMed]
- Gross MS, Phillips EA, Balen A, et al. The Malleable Implant Salvage Technique: Infection Outcomes after Mulcahy Salvage Procedure and Replacement of Infected Inflatable Penile Prosthesis with Malleable Prosthesis. J Urol 2016;195:694-7. [Crossref] [PubMed]
- Garber BB, Lim C. Inflatable Penile Prosthesis Insertion in Men with Severe Intracorporal Fibrosis. Curr Urol 2017;10:92-6. [Crossref] [PubMed]
- Trost L, Patil M, Kramer A. Critical Appraisal and Review of Management Strategies for Severe Fibrosis During Penile Implant Surgery. J Sex Med 2015;12:439-47. [Crossref] [PubMed]
- Lao M, Graydon RJ, Bieniek JM. Salvage penile prosthetic surgery utilizing temporary malleable implants. Transl Androl Urol 2017;6:S806-S812. [Crossref] [PubMed]
- Chandrapal JC, Barton GJ, Brousell S, et al. Corporal excavation and penile prosthesis placement for severe corporal fibrosis. Asvide 2019;6:214. Available online: http://www.asvide.com/watch/32899
Cite this article as: Chandrapal JC, Barton GJ, Brousell S, Lentz AC. Corporal excavation and penile prosthesis placement for severe corporal fibrosis. J Vis Surg 2019;5:67.