Jejunostomy—technique and controversies
Introduction
Malnutrition is frequently encountered is patients with esophageal cancer. It can be directly related to tumor associated dysphagia or the overall catabolic state, and can be exacerbated by the side effects of chemo-radiation treatment, mainly nausea and anorexia. Several studies have demonstrated the importance of peri-operative nutritional optimization before esophageal surgery (1). Enteral nutrition is generally preferred to parenteral nutrition, as it is associated with fewer complications rates and enhanced recovery (2). In fact, there are several negative side effects of parenteral nutrition including electrolyte disturbance, central venous access line infection, hepatic dysfunction and re-feeding syndrome. Enteral nutrition after surgery can be accomplished via oral intake or through a feeding tube.
The need for a feeding jejunostomy (FJ) before neoadjuvant therapy should be individualized depending on the level of pre-operative obstruction and weight loss (3). In fact, FJ placement has its own catheter related complications and should not be performed routinely in every patient undergoing esophagectomy as we discuss later (4,5). However, if the decision is made to place jejunostomy at the time of esophagectomy or even before, either a robotic assisted or laparoscopic approach can be utilized.
Surgical technique
Laparoscopic approach
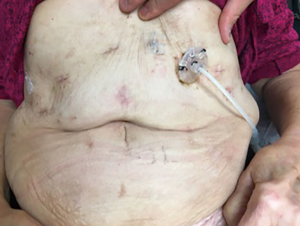
At the conclusion of the abdominal portion of a minimally invasive esophagectomy, lifting up the transverse mesocolon identifies the ligament of Treitz. About 40 cm distal to that, the jejunum is grasped. A proximal silk or Ethibond stay suture is placed, tacking the bowel to an optimal position on the abdominal wall to ensure minimal tension. The introducer needle is passed through the abdominal wall into the jejunum under direct camera vision and a wire introduced. The needle is retrieved and serial dilation is performed using a Seldinger technique. The catheter is then passed through the peel-away sheath distally into the jejunum. Care must be taken at all times not to cause an inadvertent enterotomy further distally or at the mesenteric side, with the introduction of either the needle, wire or the dilators. Verification of appropriate positioning of the tube is accomplished by injecting 20 cc of air and observation of distal bowel inflation. The balloon can then be injected with 1–2 cc of water, as not to cause any bowel obstruction. The jejunostomy insertion site can then be fashioned to the anterior abdominal wall in a stamm manner, and the tube pulled back so the balloon is only gently snug, as not to cause bowel wall necrosis. We prefer to use the Endostitch device (Covidien, CT, USA) for that portion (Figure 1). Some authors recommend the Witzel technique and prefer to tunnel the catheter proximally with several additional sutures. Finally, the tube is secured to the abdominal wall and skin using several sutures to prevent dislodgement and migration (Figure 2).

Robotic approach
If the robotic platform is used to perform the abdominal portion of the esophagectomy, it can also be utilized to perform the jejunostomy. A satisfactory portion of the jejunum is identified 40 cm distal to the ligament of Treitz as usual. The needle is then introduced into the abdomen under vision and the wire is placed into the peritoneal cavity. The tract is then serially dilated and the jejunal feeding tube is threaded into the abdominal cavity through the peel-away sheath. An enterotomy is made in the jejunum using the Maryland forceps and the feeding tube is placed inside the bowel lumen. A purse-string with 3-0 silk suture is then placed around the tube using robotic assistance. The proximal Witzel is then created using additional 3-0 silk sutures and the feeding tube—jejunostomy apparatus is stammed to the anterior abdominal wall with laparoscopic assistance as usual (Figure 3).

Outcomes of feeding jejunostomy in esophageal cancer patients
Several studies have shown that jejunostomy feeding in the early post-operative period is safe and does not negatively impact survival in esophageal cancer (8). Álvarez-Sarrado et al. for instance showed that 82.9% of patients with feeding jejunostomy reached nutritional requirements post-operatively. In addition, the use of post-operative FJ is usually short lived and more than 77% of patients do not use their tube 30 days after surgery (4). Therefore, FJ is considered a valuable bridge for nutrition and should be considered in any patient who cannot maintain adequate oral intake in the peri-operative period.
Complications and controversies
There are inherent risks and controversies associated with placement of a jejunostomy feeding tube in patients with esophageal cancer regardless of timing. Some complications related to jejunostomy placement include aspiration pneumonia, tube dislodgement leading to pneumoperitoneum and peritonitis, bowel obstruction, and abdominal wall infections. The incidence of jejunostomy related complications is noted to be as high as 38% (9). A study by Akiyama et al. showed that there was no difference in re-admission rates, hospital length of stay or pneumonia in patients who underwent jejunostomy versus those that didn’t. However, there was an increased rate of bowel obstruction (9.1%) in the jejunostomy group (5). If a bowel obstruction occurs, re-exploration is necessary and may require bowel resection. In another study, the feeding jejunostomy related overall complication rate was noted to be 51%, with 2.9% of patient’s requiring surgical re-intervention (4). A rare but often fatal complication related to early jejunostomy feeding is an entity called non-occlusive mesenteric ischemia. This presents as abdominal pain and nausea with the initiation of early post-operative enteral feeding via FJ and is associated with high osmolarity formula, extensive abdominal adhesiolysis, and pre-operative malnutrition (10).
Given the significant rate of complications that can occur with placement of feeding jejunostomy tubes in patients with esophageal cancer, many authors have advocated for early oral intake after esophagectomy. In fact, several studies have shown that such a policy is safe and is not associated with increased morbidity (11). For instance, an Enhanced Recovery After Surgery (ERAS) program in patients undergoing both minimally invasive and McKeown esophagectomy involved re-institution of a clear liquid diet by post-operative day (POD)#3 and advancement to soft diet by POD#4. The ERAS group was found to have a lower total complication rate compared to the non-ERAS group (27% vs. 44%), with earlier extubation and decreased ICU and hospital length of stay (12). Another study from Australia that implemented early oral intake within their ERAS program after esophagectomy found no difference in outcome or length of stay when patients were given clear liquids by POD#3, a soft diet by POD#7 and continued jejunostomy feeds 1-week post-discharge. In fact, they were able to show that implementing such a protocol led to greater adherence to the diet in the outpatient setting (13). On the other hand, early oral intake is not universally accepted after esophagectomy. For instance, a retrospective comparison of 3 diet regimens in 359 patients who underwent esophagectomy found that a “nil by mouth” protocol until POD#7 followed by a slow increase to a blended diet resulted in less complications and a tendency of fewer anastomotic leakages (14). Additionally, other studies have demonstrated that despite the use of enteral nutrition in the peri-operative period, these patients still have a significant decrease in body mass index (BMI), lean body and muscle mass at 6 months (15). In their retrospective review (n=151), Fenton and colleagues found that patients who benefited the most from a feeding tube were those with a BMI <18.5 (16).
In conclusion, malnutrition is frequent in patients with esophageal cancer. Placement of a feeding jejunostomy tube can be performed in a minimally invasive fashion. In view of a substantial complication rate, the decision to place a feeding tube should be individualized, and consideration made to the patients’ perioperative nutritional status and their ability to tolerate an oral intake.
Acknowledgments
Funding: This research was funded in part through the NIH/NCI Cancer Center Support Grant P30 CA006927.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Journal of Visualized Surgery for the series “Robotic Surgery for Esophageal Cancer”. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jovs.2019.03.15). The series “Robotic Surgery for Esophageal Cancer” was commissioned by the editorial office without any funding or sponsorship. AA served as the unpaid Guest Editor of the series and serves as an unpaid editorial board member of Journal of Visualized Surgery from Jul 2018 to Jun 2020. RP reports personal fees from Veran Medical, outside the submitted work. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Han H, Pan M, Tao Y, et al. Early Enteral Nutrition is Associated with Faster Post-Esophagectomy Recovery in Chinese Esophageal Cancer Patients: A Retrospective Cohort Study. Nutr Cancer 2018;70:221-8. [Crossref] [PubMed]
- Berkelmans GH, van Workum F, Weijs TJ, et al. The feeding route after esophagectomy: a review of literature. J Thorac Dis 2017;9:S785-91. [Crossref] [PubMed]
- Jenkins TK, Lopez AN, Sarosi GA, et al. Preoperative enteral access is not necessary prior to multimodality treatment of esophageal cancer. Surgery 2018;163:770-6. [Crossref] [PubMed]
- Álvarez-Sarrado E, Mingol Navarro F. Feeding Jejunostomy after esophagectomy cannot be routinely recommended. Analysis of nutritional benefits and catheter-related complications. Am J Surg 2019;217:114-20. [Crossref] [PubMed]
- Akiyama Y, Iwaya T, Endo F, et al. Evaluation of the need for routine feeding jejunostomy for enteral nutrition after esophagectomy. J Thorac Dis 2018;10:6854-62. [Crossref] [PubMed]
- Bakhos C, Patel S, Petrov R, et al. Laparoscopic jejunostomy tube placement. Asvide 2019;6:088. Available online: http://www.asvide.com/article/view/30883
- Bakhos C, Patel S, Petrov R, et al. Robotic jejunostomy tube placement. Asvide 2019;6:089. Available online: http://www.asvide.com/article/view/30884
- Klevebro F, Johar A, Lagergren J, et al. Outcomes of nutritional jejunostomy in the curative treatment of esophageal cancer. Dis Esophagus 2018; [Epub ahead of print]. [Crossref] [PubMed]
- Weijs TJ, Berkelmans GH, Nieuwenhuijzen GA, et al. Routes for early enteral nutrition after esophagectomy. A systematic review. Clin Nutr 2015;34:1-6. [Crossref] [PubMed]
- Kurita D, Fujita T, Horikiri Y, et al. Non-occlusive mesenteric ischemia associated with enteral feeding after esophagectomy for esophageal cancer: report of two cases and review of the literature. Surg Case Rep 2019;5:36. [Crossref] [PubMed]
- Berkelmans GHK, Fransen L, Weijs TJ, et al. The long-term effects of early oral feeding following minimal invasive esophagectomy. Dis Esophagus 2018;31:1-8. [Crossref] [PubMed]
- Giacopuzzi S, Weindelmayer J, Treppiedi E, et al. Enhanced recovery after surgery protocol in patients undergoing esophagectomy for cancer: a single center experience. Dis Esophagus 2017;30:1-6. [Crossref] [PubMed]
- Benton K, Thomson I, Isenring E, et al. An investigation into the nutritional status of patients receiving an Enhanced Recovery After Surgery (ERAS) protocol versus standard care following Oesophagectomy. Support Care Cancer 2018;26:2057-62. [Crossref] [PubMed]
- Eberhard KE, Achiam MP, Rolff HC, et al. Comparison of "Nil by Mouth" Versus Early Oral Intake in Three Different Diet Regimens Following Esophagectomy. World J Surg 2017;41:1575-83. [Crossref] [PubMed]
- Donohoe CL, Healy LA, Fanning M, et al. Impact of supplemental home enteral feeding postesophagectomy on nutrition, body composition, quality of life, and patient satisfaction. Dis Esophagus 2017;30:1-9. [Crossref] [PubMed]
- Fenton JR, Bergeron EJ, Coello M, et al. Feeding jejunostomy tubes placed during esophagectomy: are they necessary? Ann Thorac Surg 2011;92:504-11; discussion 511-2. [Crossref] [PubMed]
Cite this article as: Bakhos C, Patel S, Petrov R, Abbas A. Jejunostomy—technique and controversies. J Vis Surg 2019;5:33.