Management of intraoperative bleeding in the video-assisted thoracoscopic surgeries
Introduction
With the rapid growing experience, it became prominent that minimal invasive surgeries (MIS) essentially allow for expedited recover to baseline functionality (1) with less pain (2) and fewer complications (3,4). Importantly, recent large retrospective-cohort, non-inferiority study using the Society of Thoracic Surgeons General Thoracic Surgery Database suggested that MIS is not oncologically inferior to traditional thoracotomy for lung cancer (5). Similarly, superior short-term surgical results and equal oncological outcomes were achieved with video-assisted thoracoscopic (VATS) esophagectomy compared with open surgeries (6). MIS is becoming the standard of care for surgical treatment of early stage pulmonary and esophageal malignancy (1-4,7).
However, intraoperative bleeding in VATS surgeries remains the leading cause for conversion to open surgery and the most challenging risk factor. Even in the hands of experienced surgeons from high volume centers, the incidence of conversion to thoracotomy due to massive bleeding may surprisingly exceed 5% (8). In our institute, the common causes of intraoperative bleeding include dissection of calcified lymph nodes firmly adherent to the vessel, tear stress during stapler passage or firing, vascular hemostatic clips being ripped off by stapler accidentally. Logically, the proper management of intraoperative bleeding is of vital importance for VATS surgeries. Despite that immediate conversion to thoracotomy in the bleeding scenario is well advocated, a minimal invasive approach to hemorrhage control and vascular repair can be safe and effective in experienced hands.
Case presentation
We present two cases of intraoperative bleeding during thoracoscopic lobectomy and esophagectomy, demonstrating that calm maintenance of a clear thoracoscopic view, prompt tamponade of the vascular injury, precise bleeding control and suture repair of the defect are key elements for management of bleeding.
Figure 1 is a video demonstrating a uniportal thoracoscopic left upper lobectomy complicated with an accidental pulmonary arterial injury hit by an endoscopic stapler incurred while dissecting the left upper bronchus. Detailed operation procedure for uniportal lobectomy was described before (10). We maintained the thoracoscopic view, and a thoracoscopic suction was immediately placed overlying the injury for both tamponade and blood suction to get a clear vision. We then managed to clamp the vascular defect with long straight hemostatic forceps, below which a second piece of right-angled hemostatic forceps was placed. Two hemostatic forceps were alternatively released to expose the exact site of vascular defect. Consequently, the clear surgical field and adequate hemostasis allowed for a suture repair of the vascular defect with a continuous running stitch of 4-0 prolene.

Figure 2 is a video recording a minimal invasive esophagectomy complicated with an accidental esophageal nutrient artery injury incurred while dissecting the esophagus. Detailed operation procedure was described before (12). This patient suffering locally advanced esophageal carcinoma had received neoadjuvant chemoradiotherapy prior to surgery, yet the tumor mass stuck tightly with the descending aorta with obscure tissue boundary. An extended period of tamponade with gauze at the bleeding site sufficed to reach temporary hemostasis. As the tumor was massive in terms of size and volume, the view of bleeding site was obstructed and limited space was left for surgical manipulations. We therefore dissected the residual esophagus to expose the hemorrhage location, which significantly improved the visualization of defect location and facilitated the suture repair.

Conclusions
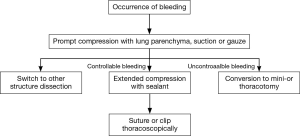
We intend to illustrate with these videos our experience for management of intraoperative bleeding in the VATS surgeries. A prompt and precise control of bleeding should be established with gentle pressure, which allows the surgeons to assess the severity of injury. Judged from their own surgical skills and experiences, the surgeons could then make a decision whether a conversion to open surgery is necessary. Under the circumstances of moderate hemorrhage, gentle tamponade with hemostasis clamp usually suffice subsequent suture repair or direct hemostasis as demonstrated in these videos. Extended period of tamponade is sometimes demanded as shown in Figure 3. There is also an option to temporarily occlude pulmonary artery trunk in case of massive bleeding or poor exposure for clamping in VATS lobectomy. The pulmonary artery trunk could be temporarily occluded by either passing a vessel loop twice around the left or right main pulmonary artery and gently applying tension to the looped suture (serving as a tourniquet) thoracoscopically (13), or directly using vascular clamp after thoracotomy. Clamping the pulmonary artery trunk within the pericardium is an option but not always necessary. We also consider it helpful to remove the residual esophagus (or lobe) to improve the visualization and operating space. Our experiences confirm the feasibility of a minimal invasive approach for intraoperative bleeding management. However, once the significant bleeding could not be controlled by compression with sealant (Fibrillar, Ethicon, San Lorenzo, Puerto Rico), gauze or vascular clamp, followed by rapid blood loss more than 800 mL, an emergent conversion to mini- or thoracotomy is recommended. Notably, compression with sealant seemed more effective when dealing with arterial injuries as compared with gauze. Hemostatic clip should be used with caution as it may cause further damage to the vessels when surgical vision is obscure. Despite the decision for conversion does vary depending on individual experience, technique and confidence of the surgeon, we have proposed an algorithm for conversion from our own experience.
Acknowledgments
Funding: This study was supported by the National Natural Science Foundation of China (81000028, 81370001, 81570031); the Key Research and Development Program of Jiangsu Province (BE2016714); the “333” Elitist Training Program, Jiangsu, China (BRA2017129); the “Six Talent Peaks” Training Program, Jiangsu, China (2014-WSN-078); the “Distinguished Medical Specialist” Program, Jiangsu, China; the “Innovative and Entrepreneurial Elite Team” Program (2016), Jiangsu, China; the research funding of Zhongshan Hospital (2016ZSLC15) and the research funding of Shanghai Hospital Development Center (SHDC12018X12).
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Seshiru Nakazawa and Kimihiro Shimizu) for the series “Emergency Response to Intraoperative Bleeding” published in Journal of Visualized Surgery. The article has undergone external peer review.
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jovs.2019.03.05). The series “Emergency Response to Intraoperative Bleeding” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Wang H, Feng M, Tan L, et al. Comparison of the short-term quality of life in patients with esophageal cancer after subtotal esophagectomy via video-assisted thoracoscopic or open surgery. Dis Esophagus 2010;23:408-14. [PubMed]
- Bendixen M, Jorgensen OD, Kronborg C, et al. Postoperative pain and quality of life after lobectomy via video-assisted thoracoscopic surgery or anterolateral thoracotomy for early stage lung cancer: a randomised controlled trial. Lancet Oncol 2016;17:836-44. [Crossref] [PubMed]
- Delaney CP, Chang E, Senagore AJ, et al. Clinical outcomes and resource utilization associated with laparoscopic and open colectomy using a large national database. Ann Surg 2008;247:819-24. [Crossref] [PubMed]
- Boffa DJ, Dhamija A, Kosinski AS, et al. Fewer complications result from a video-assisted approach to anatomic resection of clinical stage I lung cancer. J Thorac Cardiovasc Surg 2014;148:637-43. [Crossref] [PubMed]
- Boffa DJ, Kosinski AS, Furnary AP, et al. Minimally Invasive Lung Cancer Surgery Performed by Thoracic Surgeons as Effective as Thoracotomy. J Clin Oncol 2018;36:2378-85. [Crossref] [PubMed]
- Moon DH, Lee JM, Jeon JH, et al. Clinical outcomes of video-assisted thoracoscopic surgery esophagectomy for esophageal cancer: a propensity score-matched analysis. J Thorac Dis 2017;9:3005-12. [Crossref] [PubMed]
- Mathisen DJ. Is video-assisted thoracoscopic lobectomy inferior to open lobectomy oncologically? Ann Thorac Surg 2013;96:755-6. [Crossref] [PubMed]
- Samson P, Guitron J, Reed MF, et al. Predictors of conversion to thoracotomy for video-assisted thoracoscopic lobectomy: a retrospective analysis and the influence of computed tomography-based calcification assessment. J Thorac Cardiovasc Surg 2013;145:1512-8. [Crossref] [PubMed]
- Yin J, Tan L. Uniportal thoracoscopic left upper lobectomy with suture repair of pulmonary artery injury following effective bleeding control and vascular defect exposure. Asvide 2019;6:060. Available online: http://www.asvide.com/article/view/30428
- Shen Y, Wang H, Feng M, et al. Single- versus multiple-port thoracoscopic lobectomy for lung cancer: a propensity-matched studydagger. Eur J Cardiothorac Surg 2016;49:i48-53. [PubMed]
- Yin J, Tan L. Minimal invasive esophagectomy with long tamponade and subsequent suture repair of esophagus nutrient artery injury. Asvide 2019;6:061. Available online: http://www.asvide.com/article/view/30429
- Wang H, Shen Y, Feng M, et al. Outcomes, quality of life, and survival after esophagectomy for squamous cell carcinoma: A propensity score-matched comparison of operative approaches. J Thorac Cardiovasc Surg 2015;149:1006-14; discussion 1014-5.e4.
- Watanabe A, Koyanagi T, Nakashima S, et al. How to clamp the main pulmonary artery during video-assisted thoracoscopic surgery lobectomy. Eur J Cardiothorac Surg 2007;31:129-31. [Crossref] [PubMed]
Cite this article as: Yin J, Tan L. Management of intraoperative bleeding in the video-assisted thoracoscopic surgeries. J Vis Surg 2019;5:26.