The inaugural SingHealth uniportal video-assisted thoracoscopic surgery masterclass in Singapore
Introduction
Singapore Health Services (SingHealth) is Singapore’s largest group of public healthcare institutions, consisting of four public hospitals (including the largest hospital in the country, Singapore General Hospital), five national specialty centres and a network of primary care polyclinics. Multi-port video-assisted thoracoscopic surgery (VATS) has been the standard approach for most general thoracic surgical procedures performed in SingHealth since the early 2000s. However, with early evidence showing that uniportal VATS may have benefits over conventional multi-port VATS (1,2), the thoracic surgeons in SingHealth have recently begun to adopt uniportal VATS in selected cases. To further improve our expertise in this promising technique, we invited Dr. Gonzalez-Rivas to conduct the inaugural SingHealth uniportal VATS masterclass from 2–3 April 2018 at the National Heart Centre Singapore.
The National Heart Centre Singapore is a national specialty centre for cardiovascular medicine and cardiothoracic surgery in Singapore, located within the campus of the Singapore General Hospital. Each year, over 100,000 patients seek treatment at the National Heart Centre Singapore, and it serves as not only a national, but also as a regional referral centre within Southeast Asia for both cardiovascular medicine and cardiothoracic surgery. It provides comprehensive state of the art care for both cardiovascular medicine and cardiothoracic surgery, and is home to Singapore’s only heart and lung transplant program.
The uniportal VATS masterclass was conducted over a 2-day period on 2–3 April 2018. On both days, Dr. Gonzalez-Rivas delivered a lecture to the participants of the masterclass, after which he performed two live uniportal VATS surgery cases as a proctor, sharing with us the technical details and potential pitfalls at each stage of the procedure. On the first day, the lecture was on the basic principles of uniportal VATS, and the two surgical cases were a right middle lobectomy and left upper lobectomy. On the second day, after a lecture on the management of complications in uniportal VATS, two right upper lobectomies were scheduled. The second right upper lobectomy was completed via a subxiphoid approach, and also marks the first time a subxiphoid lobectomy was performed in Singapore. Details of the equipment used, procedures performed and a clinical summary of the cases that were operated on are briefly described below.
Equipment
30-degree 10 mm video thoracoscope and high definition imaging system (Olympus, Tokyo, Japan), dedicated uniportal thoracoscopic instrumentation (Scanlan International, St. Paul, Minnesota, USA), SurgiSleeve wound protector (Medtronic, Minneapolis, Minnesota, USA), EndoGIA surgical staplers (Medtronic, Minneapolis, Minnesota, USA) and LigaSure Maryland Jaw energy device (Medtronic, Minneapolis, Minnesota, USA) were used for all four cases.
Procedure
All four operations were performed under general anesthesia with double lumen endotracheal tube placement for lung isolation. All patients were placed in a lateral decubitus position, and the surgeon and camera-holding assistant both stood anterior to the patient (Figure 1).
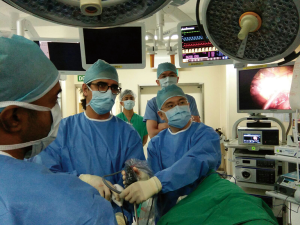
For the first three cases, a 3.5 cm incision was made at either the 4th (for the right middle and right upper lobectomy) or 5th (for the left upper lobectomy) intercostal space at the anterior axillary line. For the subxiphoid lobectomy, a 3.5 cm incision was made below the sternum, and the xiphoid process excised.
At the end of the procedure, a 28 Fr chest tube was placed through the uniportal incision for drainage. All patients were extubated in the operating room, and monitored in high dependency on the day of surgery, before being transferred to the general ward on post-operative day 1.
Case presentation
Case 1
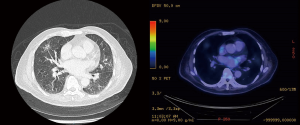
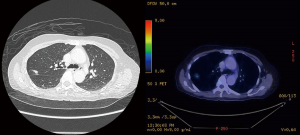
A 72-year-old male non-smoker with previous history of perforated appendicitis was found to have a 1.5 cm right middle lobe mixed density ground glass opacity on a CT thorax that was done for investigation into his chronic cough and weight loss (Figure 2). CT guided lung biopsy was performed and established the diagnosis of adenocarcinoma. Subsequent staging PET-CT showed that the right middle lobe lesion had mild FDG uptake, but he was also found to have an incidental FDG avid 8.5 cm right renal mass suspicious for a right renal cell carcinoma. After a multidisciplinary tumor board discussion, it was decided that he should undergo a staged surgical resection for what was likely to be two separate malignancies. He first underwent a laparoscopic right radical nephrectomy, and histopathology showed a pT3a clear cell renal cell carcinoma. A month after the initial operation, he then underwent a uniportal VATS right middle lobectomy and lymph node dissection that confirmed a pT1bN0 lung adenocarcinoma on histopathological examination.
Case 2
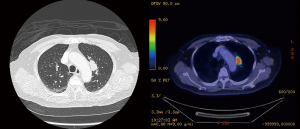
A 81-year-old male non-smoker who was discovered to have a 2.5 cm left upper lobe mass abutting the mediastinal pleura during work-up for chronic cough. A PET-CT was done, and this revealed that the left upper lobe lesion was hypermetabolic, as well as the presence of an FDG avid prevascular lymph node that was suspicious for metastasis (Figures 3,4). No other distant metastasis was seen. His imaging was discussed at a multidisciplinary tumor board, which recommended upfront lobectomy followed by adjuvant therapy, as the imaging findings were consistent with single station stage IIIA lung cancer that could be completely resected, while a CT guided biopsy or transbronchial biopsy of the lesion would be difficult in view of its location. He underwent a uniportal VATS left upper lobectomy and lymph node dissection (Figure 5), and histopathology examination established the diagnosis of pT2aN2 sarcomatoid carcinoma arising in association with adenocarcinoma. Metastatic carcinoma was seen in 1 out of 2 aortopulmonary window lymph nodes excised, but otherwise no other mediastinal lymph node was involved. 5 out of 8 lobar and 1 out of 3 intraparenchymal lymph nodes excised were also involved.

Case 3
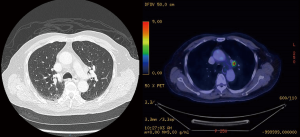
A 72-year-old female non-smoker was noted to have an incidental 1.4 cm right upper lobe nodule on chest X-ray done for investigation for chest pain. This was noted to be a spiculated lesion suggestive of malignancy on follow-up CT thorax, so a CT guided biopsy was performed, which revealed non-small cell carcinoma (Figure 6). A staging PET-CT showed no other suspicious lesions, so she underwent a uniportal VATS right upper lobectomy and lymph node dissection. Histopathological examination demonstrated a pT1bN0 sarcomatoid carcinoma arising in association with adenocarcinoma.
Case 4
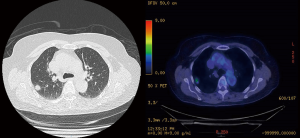
A 61-year-old female non-smoker was diagnosed with a 1.3 cm right upper lobe nodule after a CT aortogram was done for investigation of chest pain. PET-CT was done, and this lesion was found to be hypermetabolic with adjacent pleural thickening, suspicious for primary lung malignancy (Figure 7). However, the location of the lesion was thought to be difficult to biopsy by CT guided biopsy or transbronchial biopsy, so she was referred for surgical resection. The option of wedge resection versus lobectomy was discussed with the patient, and she opted for a lobectomy to avoid the potential need for a second operation in the event that final histopathology from wedge resection revealed a primary lung malignancy. Thus, she underwent a subxiphoid VATS right upper lobectomy and lymph node dissection (Figure 8), but final histopathology revealed that the lesion was a focus of necrotizing granulomatous inflammation.

Discussion
Since the development of VATS for major pulmonary resections in the early 1990s (5,6), the landscape of contemporary thoracic surgical practice has changed dramatically. While a thoracotomy or sternotomy was a strict requirement for any thoracic surgical procedure in the past, an open operation is now only needed in a minority of cases in centres with the necessary equipment and proficiency to perform VATS. Numerous studies performed worldwide have shown that VATS confers benefits in terms of less postoperative pain (7), decreased blood loss (8,9), shorter duration of stay in hospital (8,10,11) and fewer perioperative complications (9,11), with no compromise on long-term outcomes (10,12). Thus, it comes with no surprise that current guidelines in the management of lung cancer (13,14), pneumothorax (15) and empyema (16) recommend a VATS approach over an open operation whenever there is the appropriate experience and expertise available.
To this end, there has been interest amongst thoracic surgeons in reducing the number and size of incisions used in VATS to further magnify the benefits derived from the minimally invasive approach. Single incision or uniportal VATS was first described for minor thoracic surgical procedures (17-19), but interest in this technique has grown exponentially over the last few years after it was demonstrated that lobectomies can be performed safely through a single incision (20,21). It has been gradually expanded for use in progressively more complex operations, including chest wall (22), sleeve (23) and carinal resections (24), proving that it is a versatile technique in the hands of a skilled practitioner.
Much has been said about the theoretical benefits of adopting a uniportal VATS approach, including improved cosmesis, decreased surgical trauma as well as a geometrical and ergonomic advantage for the operating surgeon (25,26). In addition, there is also early clinical data which suggests that uniportal VATS may have an incremental benefit over multi-port VATS in terms of postoperative pain and length of stay in hospital (1,2). However, there are some challenges with this technique, including a higher likelihood of instrument fencing, less ability to retract the lung and additional difficulty for the assistant holding the camera suspended in mid-air, compared to conventional VATS. Advances in instrumentation including double-hinged, narrow shaft instruments (Scanlan International, St. Paul, Minnesota, USA) and variable angulated endostaplers with curved tips (Medtronic, Minneapolis, Minnesota, USA) have helped overcome some of these problems.
Even more helpful though, is the benefit of a visiting expert such as Dr. Gonzalez-Rivas to demonstrate the technique in person, share important tricks required to overcome difficult steps and troubleshoot any potential shortcomings within the local operating room setup for a uniportal VATS procedure. Moreover, having a proctor also helps guide the entire team, including the anesthetists, nurses and surgical assistants, to ensure the optimal conduct of the uniportal VATS operation. With a solid foundation on the basic principles learnt during this inaugural uniportal VATS masterclass, it is hoped that we will begin to adopt this approach for a greater number of our patients in the future, with the attendant potential benefits described previously. Moreover, it is a technique with great promise that continues to be further refined. Further improvement in post-operative pain control may be achieved by a uniportal subxiphoid VATS operation (such as the last case performed during this masterclass), as it avoids the intercostal nerves completely (27). Non-intubated thoracic operations done under locoregional anesthesia and sedation are facilitated by a uniportal approach. This may decrease perioperative morbidity by avoiding general anesthesia, which is associated with a higher risk of pneumonia and impaired cardiac function (28). Advances in thoracic surgical techniques such as these are important, to ensure that thoracic surgery remains relevant in a future where non-surgical therapeutic modalities, such as stereotactic body radiotherapy (29) and bronchoscopic ablation (30) for lung cancer and bronchoscopic lung volume reduction for emphysema (31), will challenge the status quo that surgery is the standard of care.
Acknowledgments
This masterclass was supported financially in part by Medtronic.
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Journal of Visualized Surgery for the series “Teaching Uniportal VATS”. The article has undergone external peer review.
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jovs.2018.09.01). The series “Teaching Uniportal VATS” was commissioned by the editorial office without any funding or sponsorship. DGR served as the unpaid Guest Editor of the series and serves as an unpaid associate editor-in-chief of Journal of Visualized Surgery. BHO reports non-financial support from Johnson & Johnson, non-financial support from Medtronic, non-financial support from Broncus, personal fees from AstraZeneca, outside the submitted work. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Wang L, Liu D, Lu J, et al. The feasibility and advantage of uniportal video-assisted thoracoscopic surgery (VATS) in pulmonary lobectomy. BMC Cancer 2017;17:75. [Crossref] [PubMed]
- Harris CG, James RS, Tian DH, et al. Systematic review and meta-analysis of uniportal versus multiportal video-assisted thoracoscopic lobectomy for lung cancer. Ann Cardiothorac Surg 2016;5:76-84. [Crossref] [PubMed]
- Ong BH, Gonzalez-Rivas D. Uniportal left upper lobectomy for case 2. Asvide 2018;5:754. Available online: http://www.asvide.com/article/view/27238
- Ong BH, Gonzalez-Rivas D. Subxiphoid right upper lobectomy for case 4. Asvide 2018;5:755. Available online: http://www.asvide.com/article/view/27239
- Roviaro G, Rebuffat C, Varoli F, et al. Videoendoscopic pulmonary lobectomy for cancer. Surg Laparosc Endosc 1992;2:244-7. [PubMed]
- Hazelrigg SR, Nunchuck SK, LoCicero J. Video Assisted Thoracic Surgery Study Group data. Ann Thorac Surg 1993;56:1039-43; discussion 1043-4. [Crossref] [PubMed]
- Bendixen M, Jørgensen OD, Kronborg C, et al. Postoperative pain and quality of life after lobectomy via video-assisted thoracoscopic surgery or anterolateral thoracotomy for early stage lung cancer: a randomised controlled trial. Lancet Oncol 2016;17:836-44. [Crossref] [PubMed]
- Murakawa T, Ichinose J, Hino H, et al. Long-term outcomes of open and video-assisted thoracoscopic lung lobectomy for the treatment of early stage non-small cell lung cancer are similar: a propensity-matched study. World J Surg 2015;39:1084-91. [Crossref] [PubMed]
- Boffa DJ, Dhamija A, Kosinski AS, et al. Fewer complications result from a video-assisted approach to anatomic resection of clinical stage I lung cancer. J Thorac Cardiovasc Surg 2014;148:637-43. [Crossref] [PubMed]
- Yang CF, Sun Z, Speicher PJ, et al. Use and Outcomes of Minimally Invasive Lobectomy for Stage I Non-Small Cell Lung Cancer in the National Cancer Data Base. Ann Thorac Surg 2016;101:1037-42. [Crossref] [PubMed]
- Falcoz PE, Puyraveau M, Thomas PA, et al. Video-assisted thoracoscopic surgery versus open lobectomy for primary non-small-cell lung cancer: a propensity-matched analysis of outcome from the European Society of Thoracic Surgeon database. Eur J Cardiothorac Surg 2016;49:602-9. [Crossref] [PubMed]
- Taioli E, Lee DS, Lesser M, et al. Long-term survival in video-assisted thoracoscopic lobectomy vs open lobectomy in lung-cancer patients: a meta-analysis. Eur J Cardiothorac Surg 2013;44:591-7. [Crossref] [PubMed]
- Howington JA, Blum MG, Chang AC, et al. Treatment of stage I and II non-small cell lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:e278S-313S.
- Guidelines NCCN. Non-small cell lung cancer. Available online: https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
- Baumann MH, Strange C, Heffner JE, et al. Management of spontaneous pneumothorax: an American College of Chest Physicians Delphi consensus statement. Chest 2001;119:590-602. [Crossref] [PubMed]
- Shen KR, Bribriesco A, Crabtree T, et al. The American Association for Thoracic Surgery consensus guidelines for the management of empyema. J Thorac Cardiovasc Surg 2017;153:e129-46. [Crossref] [PubMed]
- Yamamoto H, Okada M, Takada M, et al. Video-assisted thoracic surgery through a single skin incision. Arch Surg 1998;133:145-7. [Crossref] [PubMed]
- Migliore M, Deodato G. A single-trocar technique for minimally-invasive surgery of the chest. Surg Endosc 2001;15:899-901. [Crossref] [PubMed]
- Rocco G, Martin-Ucar A, Passera E. Uniportal VATS wedge pulmonary resections. Ann Thorac Surg 2004;77:726-8. [Crossref] [PubMed]
- Gonzalez-Rivas D, de la Torre M, Fernandez R, et al. Single-port video-assisted thoracoscopic left upper lobectomy. Interact Cardiovasc Thorac Surg 2011;13:539-41. [Crossref] [PubMed]
- Gonzalez-Rivas D, Paradela M, Fernandez R, et al. Uniportal video-assisted thoracoscopic lobectomy: two years of experience. Ann Thorac Surg 2013;95:426-32. [Crossref] [PubMed]
- Gonzalez-Rivas D, Xie B, Yang Y, et al. Uniportal video-assisted thoracoscopic lobectomy with en bloc chest wall resection. J Vis Surg 2015;1:7. [PubMed]
- Gonzalez-Rivas D, Fernandez R, Fieira E, et al. Uniportal video-assisted thoracoscopic bronchial sleeve lobectomy: first report. J Thorac Cardiovasc Surg 2013;145:1676-7. [Crossref] [PubMed]
- Gonzalez-Rivas D, Yang Y, Sekhniaidze D, et al. Uniportal video-assisted thoracoscopic bronchoplastic and carinal sleeve procedures. J Thorac Dis 2016;8:S210-22. [PubMed]
- Bertolaccini L, Rocco G, Viti A, et al. Geometrical characteristics of uniportal VATS. J Thorac Dis 2013;5:S214-6. [PubMed]
- Ng CS, Rocco G, Wong RH, et al. Uniportal and single-incision video-assisted thoracic surgery: the state of the art. Interact Cardiovasc Thorac Surg 2014;19:661-6. [Crossref] [PubMed]
- Song N, Zhao DP, Jiang L, et al. Subxiphoid uniportal video-assisted thoracoscopic surgery (VATS) for lobectomy: a report of 105 cases. J Thorac Dis 2016;8:S251-7. [PubMed]
- Gonzalez-Rivas D, Bonome C, Fieira E, et al. Non-intubated video-assisted thoracoscopic lung resections: the future of thoracic surgery? Eur J Cardiothorac Surg 2016;49:721-31. [Crossref] [PubMed]
- Ong BH. Surgery versus stereotactic body radiotherapy in medically operable non-small cell lung cancer. J Xiangya Med 2018;3.
- Harris K, Puchalski J, Sterman D. Recent Advances in Bronchoscopic Treatment of Peripheral Lung Cancers. Chest 2017;151:674-85. [Crossref] [PubMed]
- Miller RJ, Murgu SD. Interventional pulmonology for asthma and emphysema: bronchial thermoplasty and bronchoscopic lung volume reduction. Semin Respir Crit Care Med 2014;35:655-70. [Crossref] [PubMed]
Cite this article as: Ong BH, Gonzalez-Rivas D. The inaugural SingHealth uniportal video-assisted thoracoscopic surgery masterclass in Singapore. J Vis Surg 2018;4:200.