
Minimally invasive mitral valve surgery: tips, tricks and technique
Introduction
In minimally invasive mitral valve surgery (MIMS), the surgeon performs mitral valve surgery through a small incision (Figure 1A,B) under direct vision utilizing specialized minimally invasive instrumentation (Figure 2A,B) without a sternotomy. Both mitral valve repair and replacement can be performed minimally invasively through a small incision. A right lateral mini-thoracotomy is performed through a 4–5-cm incision in the 4th–5th intercostal space. Many reports have been published and demonstrated the benefits of MIMS (1-3). The benefits (Table 1) include faster recovery, shorter hospital stay, earlier patient ambulation, less blood transfusion requirement, lower post-op atrial fibrillation rate, lower wound infection rate and less hospitalization cost (1-3). Just as important, this approach will allow surgeons to have a better visualization of the mitral valve apparatus (mitral leaflets, annulus, papillary cords and papillary muscles), resulting in a more comprehensive operation. Mitral valve repair should always be emphasized and performed as suggested by the American Heart Association (AHA) guidelines (4,5) especially for patients with chronic primary mitral regurgitation, and hence a great direct visualization of the mitral apparatus will be extremely helpful. Different mitral repair techniques have been proposed and developed over the years (6-8). MIMS (especially for minimally invasive mitral valve repair) can have a steep learning curve and special attention needs to be paid at each step of the procedure. In this report, we attempted to provide some tips, tricks and techniques for MIMS.
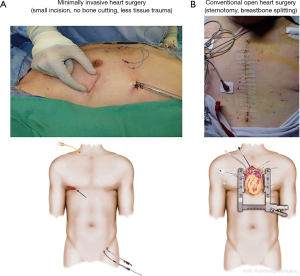
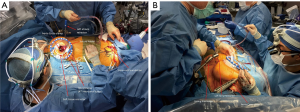
Table 1
| No breast bone splitting (no sternotomy associated morbidity) |
| Less pain with reduced trauma |
| Smaller incision |
| Better cosmesis |
| Shorter recovery time—return to normal activities quicker |
| Shorter length of hospital stay |
| Lower wound infection rate due to smaller incision |
| Lower risk of pneumonia (ambulate earlier, cough better with less pain) |
| Less blood loss/decreased blood transfusion requirement |
| Better and direct visualization of valves for better repair and replacement (better visualization than conventional open-heart surgery) |
Patient selection
In general, MIMS can be performed on most patients with mitral valve disease. Double or triple valves surgery can also be performed minimally invasively (9), but it is outside the scope of this article and will not be described here. In patient with severe peripheral vascular disease, in which peripheral cannulation for cardiopulmonary bypass is prohibited, axillary cannulation can be performed. In obese patient with mitral disease, we found it helpful to perform mitral surgery minimally invasively, and has experienced a faster recovery, earlier ambulation, shorter hospital stay and a better visualization of their mitral apparatus for those patients. For patient with prior heart surgery, MIMS can be performed with a ventricular fibrillation approach. Standard cardiac surgery preoperative screening is obtained prior to surgery. We generally do not obtain CT scan on all patients, unless there is a suspicion of peripheral vascular disease.
Surgical technique
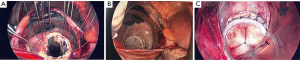
Anesthesia protocol is the same as in conventional mitral valve surgery. A Swan-Ganz catheter and radial arterial line are placed in the standard fashion. Defibrillation pad should be placed on the patient’s right shoulder and left chest. A single-lumen endotracheal tube is used since ventilation can be paused after the initiation of cardiopulmonary bypass. From our experiences, single-lung ventilation has a higher risk of unilateral pulmonary edema post-operatively. An intraoperative transesophageal echocardiogram is used for assessment of the mitral valve pathology and anatomy, which is especially important for mitral valve repair planning. We also utilize 3-D echo reconstruction. The patient is positioned with a sheet roll under the right chest, and the right arm is positioned at a 45-degree angle off to the side of the bed. The patient’s chest, abdomen and pelvis are prepped and draped in the standard surgical fashion. Cannula for cardiopulmonary bypass are generally placed in the femoral artery and vein (Figure 3A). For patient with small or calcified femoral arteries or iliac disease, the axillary artery may be used for cannulation. Seldinger technique with transesophageal echocardiogram guidance is used for femoral artery and vein cannulation. It is very important to verify the wire location in the superior vena cava before inserting the venous cannula into right atrium via the femoral vein. Fluoroscopy should be used for wire and cannula visualization if needed for confirmation if transesophageal echocardiogram cannot provide adequate images. And we generally perform limited dissection and exposure of the anterior aspect of the femoral vessels before cannulation in order to avoid lymphatics injury and lymphocele formation postoperatively. A multi-staged femoral venous cannula size 23–25 Fr should be used. And a femoral arterial cannula size 17–21 Fr should be used [body surface area (BSA) <2.0 kg/m2: 17 Fr, BSA >2.0–2.6 kg/m2: 19 Fr, BSA >2.6: 21 Fr]. The tip of the femoral venous cannula should be placed in the superior vena cava to assure adequate drainage.
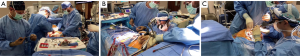
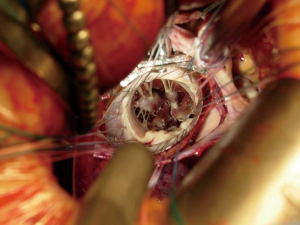
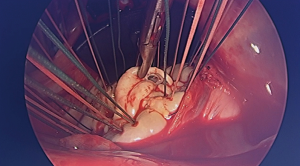
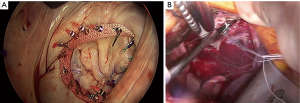
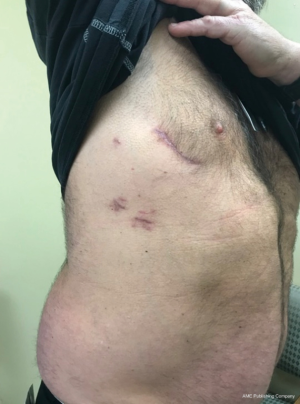
A 4–5-cm incision in the 4th or 5th intercostal space, lateral to the anterior axillary line, is made. For female patient, the incision is made laterally at the mammary crease and dissected up toward the 4th or 5th intercostal space. Plastic drape can be used to retract the breast superiorly and medially if needed. An intercostal rib retractor and soft tissue retractor are used for exposure. Extensive rib spreading is unnecessary and should be avoided in order to minimize rib fractures and postoperative pain. Cardiopulmonary bypass is initiated, and mechanical ventilation is held. Vacuum within the cardiopulmonary circuit can be used to improve blood return if needed. The pericardium is incised longitudinally about 2–3 cm above the phrenic nerve. Pericardial retraction sutures are placed and exited through the skin in order to improved exposure. We generally do not place diaphragmatic retraction suture to avoid potential liver injury, instead we use the inferior pericardial retraction sutures to retract the diaphragm inferiorly. Electrocautery and blunt dissection are used to develop a plane between the inferior aspect of the aorta and the superior aspect of the right pulmonary artery for aortic cross-clamping. A purse-string suture is placed in the mid-ascending aorta for cardioplegia/root vent cannula placement. If there is a calcified ascending aorta or in redo heart surgery, where aortic cross-clamp and anterograde cardioplegia administration are not permitted, ventricular fibrillation arrest can be utilized in the standard fashion. If there is significant aortic regurgitation, a retrograde cardioplegia catheter can be used and placed under transesophageal echocardiogram guidance through the right atrium into the coronary sinus. A chest tube incision is made two to three rib spaces below the operative incision, and a cardiotomy suction is placed. The Waterson’s groove is dissected accordingly to expose the left atrium. Extensive Waterson’s groove dissection is not needed unlike in sternotomy approach. A specialty-designed flexible aortic cross-clamp is used and placed in the previously developed plane via the same incision. Antegrade cardioplegia is given. We use modified Del Nido cardioplegia solution. A left lateral atriotomy is performed via the Waterson’s groove to expose the mitral valve. An atrial lift retractor blade is placed into the left atrium. The post of the left atrial lift retractor is inserted anteriorly into the chest lateral to the internal mammary artery (Figure 3B). A spinal needle can be placed first to identify the needle position of the post placement. Special attention is necessary in order to avoid injuring the right internal mammary artery when placing the retractor post. Visualization of the right internal mammary artery should be obtained either directly or with a thoracoscope. With proper placement of the left atrial retractor, perfect and direct visualization of the mitral valve apparatus can be obtained (Figures 3C,4). Once optimal visualization is obtained, the mitral valve is then inspected, and replacement (Figure 5A,B,C) or repair (Figures 6,7A,B) can be performed as needed. For repair, we generally place annular sutures first (Figure 6), which will further improve the visualization of the mitral apparatus. Leaflets resections, artificial chord placement or chordal translocation and mitral annuloplasty (Figures 6,7A,B) can be easily performed under direct vision. For patient with atrial fibrillation, the left atrium appendage ostium can be closed from within the left atrium using 4-0 or 5-0 prolene sutures. It is recommended to place at least two layers of closure to prevent recanalization or reopening of the appendage in the future, in which we have not experienced with this closure. A MAZE procedure will also be performed through the same incision in patients with atrial fibrillation using both radiofrequency ablation and cryoablation, which is outside the scope of this report and will not be described here. Upon completion of the mitral repair or replacement, the atriotomy is closed with 4-0 prolene suture in a running two-layer fashion. A right ventricular pacing wire is placed at this time, since it will be difficult to place after the aortic cross-clamp is removed. The patient is then place in the Trendelenburg position, and the aortic cross-clamp is removed. The aortic root vent is used for deairing. Cardiopulmonary bypass is weaned. Protamine is given and decannulation is performed in the standard fashion. For patient with femoral arterial cannulation, it is always important to examine distal pulses for confirmation of patency. At least two chest tubes should be placed with one placed within the pericardial space posteriorly and one in the right pleural space. The groin and chest are closed in the standard fashion (Figure 8).
Conclusions
MIMS is mitral valve surgery performed through a small incision under direct visualization without a sternotomy or rib division. Numerous reports have been published and proven its benefits. MIMS is surgically demanding, and cautions and attention to details are needed to perform the procedure safely and effectively. With the knowledge of the tips, tricks and technique of the procedure, patients will benefit greatly from the success of the operation.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Journal of Visualized Surgery for the series “Minimally Invasive Cardiac Surgery”. The article has undergone external peer review.
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jovs.2018.08.08). The series “Minimally Invasive Cardiac Surgery” was commissioned by the editorial office without any funding or sponsorship. AC served as the unpaid Guest Editor of the series and serves as an unpaid editorial board member of Journal of Visualized Surgery from Jun 2017 to May 2019. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Individual informed consent was obtained from the patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Galloway AC, Schwartz CF, Ribakove GH, et al. A decade of minimally invasive mitral repair: long-term outcomes. Ann Thorac Surg 2009;88:1180-4. [Crossref] [PubMed]
- Iribarne A, Easterwood R, Russo MJ, et al. Comparative effectiveness of minimally invasive versus traditional sternotomy mitral valve surgery in elderly patients. J Thorac Cardiovasc Surg 2012;143:S86-90. [Crossref] [PubMed]
- Moscarelli M, Fattouch K, Casula R, et al. What Is the Role of Minimally Invasive Mitral Valve Surgery in High-Risk Patients? A Meta-Analysis of Observational Studies. Ann Thorac Surg 2016;101:981-9. [Crossref] [PubMed]
- Nishimura RA, Otto CM, Bonow RO, et al. 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Thorac Cardiovasc Surg 2014;148:e1-132. Erratum in: J Thorac Cardiovasc Surg 2014;64:1763. Dosage error in article text. [Crossref] [PubMed]
- Nishimura RA, Otto CM, Bonow RO, et al. 2017 AHA/ACC Focused Update of the 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2017;135:e1159-95. [Crossref] [PubMed]
- Cheng A, Nguyen TC, Malinowski M, et al. Undersized mitral annuloplasty inhibits left ventricular basal wall thickening but does not affect equatorial wall cardiac strains. J Heart Valve Dis 2007;16:349-58. [PubMed]
- Nguyen TC, Cheng A, Tibayan FA, et al. Septal-lateral annnular cinching perturbs basal left ventricular transmural strains. Eur J Cardiothorac Surg 2007;31:423-9. [Crossref] [PubMed]
- Langer F, Rodriguez F, Cheng A, et al. Posterior mitral leaflet extension: an adjunctive repair option for ischemic mitral regurgitation?. J Thorac Cardiovasc Surg 2006;131:868-77. [Crossref] [PubMed]
- Santana O, Xydas S, Williams RF, et al. Outcomes of minimally invasive double valve surgery. J Thorac Dis 2017;9:S602-6. [Crossref] [PubMed]
Cite this article as: Cheng A, Ramsey AM. Minimally invasive mitral valve surgery: tips, tricks and technique. J Vis Surg 2018;4:182.


