Uniportal video-assisted anterior upper lobe anatomic segmentectomy S3: a technical description
Introduction
Video-assisted anatomic pulmonary segmental resection (segmentectomy), is the procedure of choice for patients diagnosed with stage I non-small cell lung cancer (NSCLC), but who are unfit for lobectomy (provided that a margin at least equal to the diameter of the tumor can be achieved), and when treating peripheral nodules that have at least one of the following characteristics: (I) pure adenocarcinoma in situ (AIS); (II) ≥50% (pure or pure predominant) ground glass appearance on CT and (III) a long doubling time, exceeding 400 days, confirmed following surveillance (1). Segmentectomy is also indicated for the surgical removal of lung metastasis when a wedge resection cannot be performed (2). The endorsement of the procedure, undoubtfully, reflects its efficiency, measured in long term survival, recurrence free survival, postoperative quality of life, and postoperative preserved pulmonary function (3-6), given of course that the aforementioned indications are implemented during the decision-making process and that oncological principles are not compromised.
The cornerstone of a successful segmentectomy is of course the preoperative identification of the target segment and the bronchovascular structures that have to be transected. The evolution of the imaging technology combined with planning software, allows the regeneration of detailed 3D images (7,8). These images demonstrate in detail the bronchovascular anatomy of the lung and aid the surgeon not only to identify the affected segment, but also to plan the resection, avoiding any pitfalls resulting from anatomical variations. Detailed 3D images can also be printed, delineating in an even more visual way the complex and diverse segmental anatomy (9).
The purpose of this video article is to demonstrate in a detailed way the resection of the anterior (S3) segment of the right upper and left upper lobe via the uniportal video-assisted thoracoscopic surgery (VATS) technique.
Branching arterial, venous and bronchial patterns of the right upper lobe
A thorough knowledge of the bronchovascular segmental anatomy and its variations is required when performing a segmentectomy.
The most common arterial branching pattern for the right upper lobe is that in which the arterial supply of the apical (S1) and anterior segments (S3) originate from the truncus anterior (also referred to as truncus superior), whereas the posterior segment (S2) is most often supplied by the posterior ascending artery (subsegment S2b) and the recurrent artery (subsegment S2a). The posterior ascending artery takes off separately from the interlobar artery, whereas the recurrent artery originates from the truncus anterior (10).
The branching pattern of the right upper pulmonary vein has three main variations. The most common, is that in which an apical (V1) and a central (V2) vein are present. The anterior segment is drained into these veins, via tributaries that arise early in their course (10).
The branching pattern of the right upper bronchus to its segmental bronchi is usually more consistent. In about 60% of the cases, it trifurcates to an apical (S1), posterior (S2) and anterior (S3) segmental bronchus, while in the remaining cases the apical and the posterior bronchi arise from a common trunk (11,12). The segmental bronchus, usually, lies posterior to the according segmental artery.
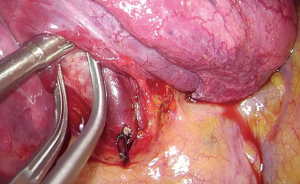
Anterior (S3) segmentectomy of the right upper lobe (Figure 1)

The case presented here has a typical vein anatomy. Both central and apical veins are present. The anterior segment drains via two separate venous branches into them. Also, the middle lobe vein is identified as the most caudal tributary of the right upper pulmonary vein. The arterial pattern is also typical as both A3a and A3b originate from the truncus anterior. Lymph node sampling or dissection is performed according to existing guidelines.
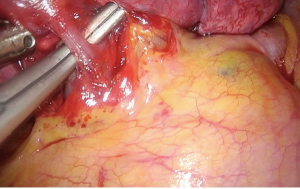
The chest is usually entered via the fourth intercostal space through a 3-cm incision at the level between the anterior and midaxillary line. The dissection of the hilum and the fissure are facilitated when the incision is aligned to the fissure. As a general rule of thumb, this is accomplished when the extension of the incision crosses the tip of the scapula, after the patient has been properly positioned. A wound protector may be used as a means to retract the soft tissue from the intercostal space, thus protecting the camera from being repeatedly stained and of course to prevent seeding of cancer cells during retrieval of the specimen. The wound protector can be particularly helpful when operating on obese patients. However, the maneuverability of the instruments can be impaired. A useful trick, when operating without a wound protector, is to retract the soft tissues using a stay suture fixed on the drapes.
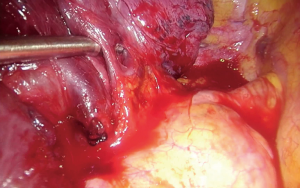
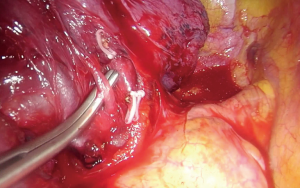
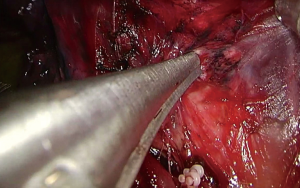
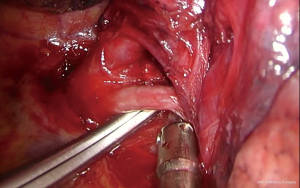
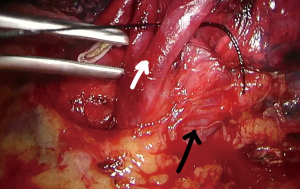
Upon entering the chest, any adhesions should be taken down in order to facilitate the maneuverability of the lobe and the culprit lesion identified and palpated if possible. With the 30° camera placed at the posterior part of the incision the upper lobe is grasped with a lung grasper and retracted posteriorly, and upwards. This maneuver exposes the hilum and the surgeon can use the sucker to acquire a tactile sensation of the structures and their anatomy. Following identification of the middle lobe vein, blunt and sharp dissection is carried out in a bimanual fashion, using the sucker in one hand and an energy device (preferably) in the other, until the interlobar artery is identified (Figure 2). A dissector (wright angle or 45° dissector) is placed upon the artery with its tip pointing at the confluence of the fissures and is used to create a tunnel along the interlobar artery. Previous dissection at the confluence can facilitate the procedure, especially when dealing with a thick fissure. While attempting this maneuver, the surgeon should handle the lung grasper in order to retract the upper lobe anteriorly and maneuver it accordingly. The presence of lymph nodes may block the passage of the dissector, requiring removing them. Once the tunnel is created the horizontal fissure is dissected using a stapling device, taking care not to compromise the middle lobe vein. Again, in case of a thick fissure a stapler can be fired before the creation of the tunnel, provided that the interlobar artery has been identified. In such a case, the anvil of the stapler is placed parallel to the middle lobe vein, while pointing at the interlobar artery. The dissection of the horizontal fissure provides a better view and understanding of the vein anatomy. Pulling the lung posteriorly and upwards again helps identifying the V3a (Figure 3), branching early from the central vein. The vein is dissected free, ligated and severed (clips, energy devices and a suture may be used). Maintaining the same traction and dissecting along the central vein leads to the identification of A3 as it crosses over the central vein and posterior to the apical vein. At this point the V3b is identified branching from the apical vein, just anterior to the A3 (Figure 4). It is dissected free, ligated and severed. It should be noted though, that usually the V3b originates from the central vein proximal to the origin of V3a. A3 is divided following the division of V3b (Figure 5). B3 lies posterior to A3 and is exposed and treated right after the division of A3 (Figure 6). The division of the intersegmental plane is the last step of the procedure. Typically, it involves the implementation of the inflation-deflation technique. However, in the presented video, the intersegmental plane is dissected based on the knowledge of the segmental spatial anatomy, with the use of a stapling device. When positioning the device and before firing the stapler, the surgeon should make sure that the stumps of the structures previously divided are incorporated into the resected segment. The specimen is removed from the chest, test for air leak and repairs (if needed) are conducted as usual and a chest tube is placed anteriorly. The lung is inflated taking care to resolve atelectatic regions before closing the incision.

Branching arterial, venous and bronchial patterns of the left upper lobe
The arterial branching pattern for the left upper lobe also exhibits a lot of variations. The anterior segment (S3), is usually supplied by a trunk, arising from the mediastinal side of the left pulmonary artery (mediastinal type). The lingular arterial supply, most often, originates from the interlobar artery (interlobar type) either as a common trunk (before bifurcating) branching distally to the take-off of A6, or as two separate branches, each corresponding to one of the two lingular subsegments. In some cases, however, the lingular artery branches from the mediastinal side of the main pulmonary artery (mediastinal type). The third branching pattern for the lingula is the least common and is a combination of the interlobar and mediastinal type.
Segments S1+2 and S3 almost always drain to the superior pulmonary vein via two separate veins (apical type). Rarely they form a common trunk, before ending up to the superior pulmonary vein. The most inferior tributary of the superior pulmonary vein is the lingular vein.
The left upper bronchus bifurcates to a bronchus for the apicoposterior (S1+2) and the anterior segment (S3) and a bronchus for the lingula. The bronchus for the S1+2 and S3 present several variations, most common of which (almost 50%) is a separate segmental bronchus for each one of the segments (14).
Anterior (S3) segmentectomy of the left upper lobe (Figure 7)

In the case presented, A3 arises from the mediastinal side of the main pulmonary artery and the segment is drained via a separate vein to the superior pulmonary vein. The dissection of the intersegmental plane is carried out based on the surgeon’s knowledge of the spatial segmental anatomy, without inflating and deflating the lung.
The chest is usually entered via the fifth intercostal space through a 3-cm incision at the level between the anterior and midaxillary line. The fourth intercostal space can also be used, since access to the fissure is not usually required for a left anterior segmentectomy.
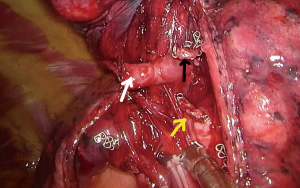
With the 30-degree high definition camera placed at the posterior part of the incision, the upper lobe is grasped with a lung grasper and with a rotating maneuver of the arm, the lung is retracted inferiorly and posteriorly, and the hilum is exposed. Again, gentle palpation of the hilar structures allows the surgeon to visualize the anatomy while performing a blunt dissection of the fatty tissue covering the hilum. With the sucker in one hand and an energy device in the other (bimanual instrumentation), dissection is carried out along the superior border of the superior pulmonary vein, starting from the level of the pericardium, until the take-off of A3 is recognized. The A3 takes off from the mediastinal side of the main pulmonary artery separately (Figure 8). Sometimes the dissection around A3 can be challenging due its proximity to the mediastinum and the presence of lymph nodes. Proper adjustment of the traction can facilitate the dissection. Once dissected free, the A3 is ligated and divided. The vein tributaries for the S1+2, S3 and the lingula are identified (Figure 9). The V3, usually the middle tributary, is dissected free, ligated and divided. Sharp dissection is resumed until the B3 is identified, lying behind V3 (Figure 10). Following the division of B3 the intersegmental plane is dissected with a stapling device. A segmentectomy can only be technically intact when the distal stumps of the severed structures are incorporated into the resected segment (Figure 11). This can sometimes be facilitated by grasping and pulling the bronchial stump up. The specimen is removed from the chest, the parenchyma is tested for the presence of air leak, and a chest tube is placed anteriorly before closing up the patient.
Comments
The merits of a segmentectomy, when utilized in accordance to published guidelines, combine the oncological efficacy of a radical operation with the advantage of sparing lung parenchyma. The increasing rate of detection of early stage cancerous lesions, resulting from lung cancer awareness and the advancements in imaging technology, has led to the identification of a large population on which a segmentectomy would be equally radical, safe and efficient compared to a lobectomy. Thorough knowledge of the segmental anatomy and its variations and correct preoperative and intraoperative identification of the target segment are the cornerstone for a technically intact and thus oncologically efficient procedure.
The preoperative identification of the target segment, as mentioned above, is based on the 3D images obtained from thin-sliced CT scans and 3D printing of these images. The intraoperative identification of the intersegmental borders of the target segment and its bronchovascular bundle can be challenging, especially when resecting only one segment. A series of strategies have been developed to tackle this issue. The inflation-deflation technique performed after the division of the segmental bronchus is the most commonly used, however due to collateral ventilation the demarcation of the intersegmental border can sometimes be poor. Others advocate the inflation of the lung before the division of the bronchus, so that the air trapped in the target segment may delineate its borders when neighboring borders have collapsed, or the selective insufflation of air into the segment (16-18). Several teams have also described the usefulness of indocyanine green in the identification of the intersegmental border (19). In the cases presented in this article the division of the intersegmental plane was based on the surgeon’s knowledge of the spatial anatomy of the segment, after inflation and deflation test, placing the stapler in between the proximal and distal bronchial segmental stump. The uniportal VATS technique provides a direct visualization to the hilum and enables a bimanual instrumentation to the surgeon working in s sagittal plane, which is especially useful for anterior (S3) segmentectomy (20,21).
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Alessandro Brunelli) for the series “Uncommon Segmentectomies” published in Journal of Visualized Surgery. The article has undergone external peer review.
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jovs.2018.06.17). The series “Uncommon Segmentectomies” was commissioned by the editorial office without any funding or sponsorship. DGR serves as an unpaid associate editor-in-chief of Journal of Visualized Surgery. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- NCCN Clinical Practice Guidelines in Oncology. Non-Small Cell Lung Cancer. Version 3.2018. Available online: www.nccn.org
- Berry MF. Role of segmentectomy for pulmonary metastases. Ann Cardiothorac Surg 2014;3:176-82. [PubMed]
- Bedetti B, Bertolaccini L, Rocco R, et al. Segmentectomy versus lobectomy for stage I non-small cell lung cancer: a systematic review and meta-analysis. J Thorac Dis 2017;9:1615-23. [Crossref] [PubMed]
- Qu X, Wang K, Zhang T, et al. Long-term outcomes of stage I NSCLC (≤3 cm) patients following segmentectomy are equivalent to lobectomy under analogous extent of lymph node removal: a PSM based analysis. J Thorac Dis 2017;9:4561-73. [Crossref] [PubMed]
- Martin-Ucar AE, Nakas A, Pilling JE, et al. A case-matched study of anatomical segmentectomy versus lobectomy for stage I lung cancer in high-risk patients. Eur J Cardiothorac Surg 2005;27:675-9. [Crossref] [PubMed]
- Harada H, Okada M, Sakamoto T, et al. Functional advantage after radical segmentectomy versus lobectomy for lung cancer. Ann Thorac Surg 2005;80:2041-5. [Crossref] [PubMed]
- Abdelsattar ZM, Blackmon SH. Using novel technology to augment complex video-assisted thoracoscopic single basilar segmentectomy. J Thorac Dis 2018;10:S1168-S1178. [Crossref] [PubMed]
- Shimizu K, Nakazawa S, Nagashima T, et al. 3D-CT anatomy for VATS segmentectomy. J Vis Surg 2017;3:88. [Crossref] [PubMed]
- Oizumi H, Kato H, Endoh M, et al. Techniques to define segmental anatomy during segmentectomy. Ann Cardiothorac Surg 2014;3:170-5. [PubMed]
- Nomori H, Okada M. Illustrated Anatomical Segmentectomy for Lung Cancer. Japan: Springer, 2012:25.
- Boyden EA, Scannell JG. An analysis of variations in the bronchovascular pattern of the right upper lobe of 50 lungs. Am J Anat 1948;82:27-73. [Crossref] [PubMed]
- Savithri K, Vidhya J. Dharani. Branching Pattern of Bronchial Tree in Both Lungs. IOSR Journal of Dental and Medical Sciences 2015;14:1-5.
- Soultanis KM, Gonzalez-Rivas D. Uniportal VATS anatomic right upper anterior segmentectomy (S3). Asvide 2018;5:609. Available online: http://www.asvide.com/article/view/25798
- Argueta AJO, Cañas SRR, Zhu Y, et al. Bilateral uniportal video-assisted thoracic surgery segmentectomy S3: an unusual case. J Vis Surg 2017;3:168. [Crossref] [PubMed]
- Soultanis KM, Gonzalez-Rivas D. Uniportal VATS anatomic left upper anterior segmentectomy (S3). Asvide 2018;5:610. Available online: http://www.asvide.com/article/view/25799
- Tsubota N. An improved method for distinguishing the intersegmental plane of the lung. Surg Today 2000;30:963-4. [Crossref] [PubMed]
- Okada M, Mimura T, Ikegaki J, et al. A novel video-assisted anatomic segmentectomy technique: selective segmental inflation via bronchofiberoptic jet followed by cautery cutting. J Thorac Cardiovasc Surg 2007;133:753-8. [Crossref] [PubMed]
- Kamiyoshihara M, Kakegawa S, Ibe T, et al. Butterfly-needle video-assisted thoracoscopic segmentectomy: a retrospective review and technique in detail. Innovations (Phila) 2009;4:326-30. [Crossref] [PubMed]
- Abdelsattar ZM, Blackmon SH. Using novel technology to augment complex video-assisted thoracoscopic single basilar segmentectomy. J Thorac Dis 2018;10:S1168-S1178. [Crossref] [PubMed]
- Gonzalez-Rivas D, Mendez L, Delgado M, et al. Uniportal video-assisted thoracoscopic anatomic segmentectomy. J Thorac Dis 2013;5:S226-33. [PubMed]
- González-Rivas D, Lirio F, Sesma J. Uniportal anatomic combined unusual segmentectomies. J Vis Surg 2017;3:91. [Crossref] [PubMed]
Cite this article as: Soultanis KM, Gonzalez-Rivas D. Uniportal video-assisted anterior upper lobe anatomic segmentectomy S3: a technical description. J Vis Surg 2018;4:140.