Robotic internal mammary lymphadenectomy: another possible minimally invasive approach to sampling lymph nodes in breast cancer patients
Introduction
Internal mammary (IM) lymphadenopathy may develop in breast cancer patients with silicone implants. It is typically asymptomatic, and it is usually diagnosed on imaging performed for follow-up. Further clinical evaluation with PET scan can confirm the presence of internal mammary adenopathy, but even if PET-positive lesions have a high probability to be malignant, false-positive uptake is not uncommon. Differential diagnosis includes infections, inflammations and granulomatous deposit. Silicon deposit in regional lymph nodes is uncommon, although well documented for axillary adenopathy in a woman with silicone implants following mastectomy for cancer.
Internal mammary lymph nodes biopsy is frequently performed percutaneously. However, the proximity to the mammary vessels may preclude the safety of the procedure. Many authors have reported the efficacy and safety of internal mammary nodes sampling performed by VATS.
Robotic manipulation of the internal mammary pedicle, previously described for arterial harvesting for the coronary bypass procedure, can be an alternative and safe option to perform a minimally invasive lymph node dissection in case of suspicious adenopathy.
We report a case of a lady with a previous history of breast cancer that underwent robotic lymph nodes dissection for internal mammary adenopathy.
Case presentation
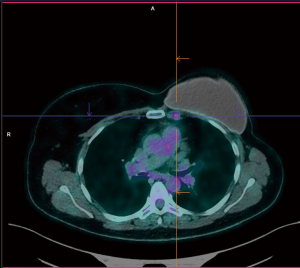
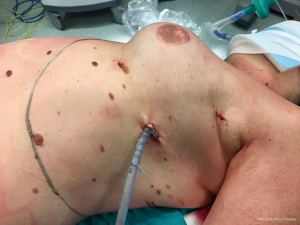
A 47-year-old female was admitted to our institution with a diagnosis of adenopathy of the left internal mammary chain. Five years earlier patient underwent left mastectomy and systemic chemotherapy followed by silicone prosthetic implants, for breast cancer. During routine follow-up patient performed CT scan and subsequent PET scan that revealed the presence of a left internal mammary adenopathy; the most significant lymph nodes measured 12 mm × 18 mm, with an SUV of 2.7 (Figure 1). As recurrent malignancy was suspected, the oncologist recommended performing tissue biopsy. A percutaneous ultrasound-guided biopsy was considered unsafe as lymph nodes were located too deep in the chest surrounded by the internal mammary vessels. We decided to proceed with a surgical biopsy performing lymph node dissection by the robotic approach. The procedure was performed in general anaesthesia with double lumen intubation to obtain one-lung ventilation. The patient was placed in horizontal semi-oblique position with the left chest elevated (Figure 2). The procedure was performed with the Si da Vinci Surgical System (Intuitive, Surgical Sunnyvale, CA, USA). We started by placing the camera port in a fifth intercostal plane at the level of the mid-axillary line. Under view guidance, the two-operative port was placed in third intercostal space at the level of the anterior axillary line and in the 5th intercostal space 4 cm lateral to the parasternal line. We then started insufflating carbon dioxide (CO2) with an 8 L/min flow and 8–10 mmHg pressure. CO2 insufflation helps to collapse the lung and to facilitate dissection of the fat tissue, efficiently creating a pneumomediastinum. We completed the set up docking the da Vinci Si. Dissection was conducted using only two robotic arms: the bipolar forceps and the cautery hook. After incising the left mediastinal pleura, we identified the internal mammary artery and vein, we dissected the fat tissue, and all visible lymph nodes were removed. At the end of the procedure, a 24 Fr drainage tube was placed in the pleural space, and the lung was inflated under direct view. Overall the operative time was 90 minutes (docking/undocking time overall 25 minutes). The patient recovered well, and no complication occurred. In post-operative day two patients were discharged. Final pathological histology revealed reactive adenopathy with granulomatous reaction. The patient was referred to the Oncologist to proceed with the routine follow up (Figure 3).

Discussion
The internal mammary lymph nodes are the second most crucial nodal basin in breast cancer, and metastasis located at this level is well known as a poor prognostic indicator. However, in patients presenting with internal mammary adenopathy with PET-positive uptake tissue sampling should be recommended to confirm the suspect of recurrent malignancy and differential diagnosis with inflammatory or granulomatous reaction should be considered in women who undergone mastoplasty with silicone implants (2,3).
Rapture or leakage of silicone breast prosthesis can cause lymphatic spread which involves more frequently axillary lymph nodes while spread to the IM nodes is rare. However, in literature have been reported numerous cases of post-mastectomy patients with silicone induced IM adenopathy mimicking cancer recurrence on PET scan. This challenging diagnosis is not so rare. Therefore histological diagnosis is paramount as a prognostic factor and for planning adjuvant chemoradiotherapy.
Biopsy of the IM lymph nodes can usually be performed successfully percutaneously (ultrasound-guided needle aspiration biopsy) however in patients with elevated BMI or thick muscle; lymph nodes cannot be easily identified. Moreover, pleural breach, vascular injuries and pneumothorax are the most commonly reported complications (4).
Numerous authors have reported and proved the safety and efficacy of the VATS approach in patients with IM lymphadenopathy. VATS carry minimal surgical trauma with a meagre rate of complications. It is a "simple” and rapid procedure that allows achieving an accurate diagnosis.
The rule of the robotic approach for a diagnostic procedure such as IM nodes dissection has not been investigated yet. Robotic manipulation of the internal mammary pedicle seems safe and allows meticulous dissection with complete removal of all the lymph nodes and fat tissue along the vessels bundle. The Robotic approach can be considered an alternative minimally invasive approach for patient with suspected metastatic IM lymphadenopathy, however prospective studies and more extensive series are needed to identify possible advantages over VATS (5,6).
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: A written informed consent was signed by the patient and was attached in the clinical dossier. Institutional review board approved this study.
References
- Pardolesi A, Bertolaccini L, Brandolini J, et al. Robotic left internal mammary lymph nodes dissection. Asvide 2018;5:389. Available online: http://www.asvide.com/article/view/24129
- Ganau S, Tortajada L, Rodriguez X, et al. Silicone lymphadenopathy: an unusual cause of internal mammary lymph node enlargement. Breast J 2008;14:502-3. [Crossref] [PubMed]
- Ho L, Wassef H, Seto J. FDG PET/CT imaging in granulomatous changes secondary to breast silicone injection. Clin Radiol 2010;65:659-61. [Crossref] [PubMed]
- Fornage BD, Dogan BE, Sneige N, et al. Ultrasound-guided fine-needle aspiration biopsy of internal mammary nodes: technique and preliminary results in breast cancer patients. AJR Am J Roentgenol 2014;203:W213-20. [Crossref] [PubMed]
- Bucerius J, Metz S, Walther T, et al. Endoscopic internal thoracic artery dissection leads to significant reduction of pain after minimally invasive direct coronary artery bypass graft surgery. Ann Thorac Surg 2002;73:1180-4. [Crossref] [PubMed]
- Bolotin G, Scott WW, Austin TC, et al. Robotic skeletonizing of the internal thoracic artery: is it safe? Ann Thorac Surg 2004;77:1262-5. [Crossref] [PubMed]
Cite this article as: Pardolesi A, Bertolaccini L, Brandolini J, Solli P. Robotic internal mammary lymphadenectomy: another possible minimally invasive approach to sampling lymph nodes in breast cancer patients. J Vis Surg 2018;4:71.