Uniportal thoracoscopic resection of intralobar and extralobar pulmonary sequestration
Introduction
Pulmonary sequestration (PS) is an uncommon congenital malformation of the lower respiratory tract in which a nonfunctional part of the lung parenchyma lacks its normal communication with the tracheobronchial tree and receives an anomalous arterial blood supply from the systemic circulation first described by Pryce in the 1946 (1). Generally the arterial blood comes from a single anomalous branch of the thoracic or less frequently are multiple or from the abdominal aorta. Generally, the venous connection is normal, and in very rare cases there is an anomalous connection with the bronchial tree (2,3). The majority of PS is classified into two main categories: intralobar sequestration (ILS) and extralobar sequestration (ELS). ILS is a lesion that lies within the normal lobe and lacks separate visceral pleura, while ELS is a formation located outside the lung separated from the normal lobe with a layer of visceral pleural. The embryologic basis of PS is still unclear; one explanation is an abnormality of lung bud formation occurring early prior the separation of the aortic and pulmonary circulation (2). The clinical presentation depends from size, type and location of the PS. Surgical resection is generally indicated in all cases of PS to resolve symptoms and prevent future complications (4,5). In the past years, surgery for PS were performed through thoracotomy, more recently multiport video-assisted thoracoscopic surgery (VATS) became the preferred approach in many centers (6-8). We report our experience in the treatment of ILS and ELS using the uniportal VATS technique.
Case presentation
Patients and preoperative work-up
The preoperative work up for PS in our institution includes a complete cardiological evaluation with transthoracic echocardiography, total body angio-CT, spirometry test and flexible bronchoscopy. If necessary, magnetic resonance imaging (MRI) and/or selective angiography should be performed. In both reported cases MRI and angiography were not necessary because the angio-CT scan images were considered optimal for the identification of the aberrant vessels and surgical planning.
Patient 1 (Figure 1)
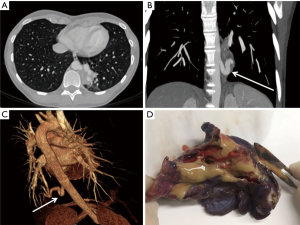
A 26-year-old female was admitted to our hospital because of left lower lobe pneumonia. At the clinical history the patient reports recurrent episodes of respiratory infection since childhood, much more frequent in the last 5 years. A computed tomography (CT) scan of the thorax was performed documenting the presence of a lesion in the basal segments of left lower lobe, with a diameter about 8.6 cm × 4.4 cm × 3.2 cm (Figure 1A). The angio-CT-scan showed the presence of an aberrant arterial branch of the descending thoracic aorta measuring 9 mm in diameter connected to the lesion (Figure 1B,C). Findings matched with intra-lobar sequestration. Further pre-operative investigations comprising bronchoscopy, pulmonary function test and echocardiography were strictly normal. Patient was recommended for surgery and she was referred to our department.
Patient 2 (Figure 2)
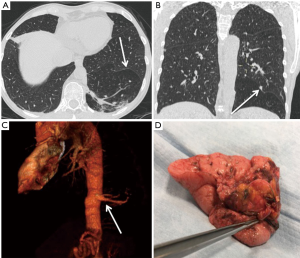
The patient was a 56-year-old woman with history recurrent respiratory infection and a recent endocarditis of the aortic valve with brain embolisms. After treatment of the endocarditis, the following cardiological, vascular and neurological evaluations were negative. Thus she underwent total body angio-CT-scan which showed an anomalous area of left lower lobe parenchyma separated with the normal one by an accessory fissure (Figure 2A,B) with two separate feeding arteries arising from the descending thoracic aorta suggestive for extralobar lung sequestration (Figure 2C). Further, pre-operative investigations comprising bronchoscopy, pulmonary function test and echocardiography were strictly normal. The patient was recommended for surgery and she was referred to our department.
Pre-operative preparation
Routine laboratory tests, chest X-ray, electrocardiography (EKG).
Equipment preference card: all surgical instruments used were exactly the same for both patients.
- Thirty-degree, 10 mm thoracoscope;
- A wound protector (Alexis® S, Applied Medical, Rancho Santa Margarita, CA, USA);
- Endoscopic graspers;
- A cautery hook (Covidien Products—Medtronic, Minneapolis, MN, USA);
- Harmonic scalpel 36 cm (Harmonic ACE®+, Ethicon Inc., Cincinnati, OH, USA);
- Mechanical endostapler (Endo GIA™ Ultra Universal Standard, Covidien Products—Medtronic, Minneapolis, MN, USA);
- Endoscopic sponge stick (Ethicon Inc., Cincinnati, OH, USA);
- Digital draining system (Thopaz™, Medela Italia Srl,).
Patients positioning
A double-lumen endotracheal tube is inserted, then, the patients are placed in a lateral decubitus on the table next to its anterior edge with the arm in the swimming position. The table is arched to open the intercostal space. The first surgeon stands on the abdominal side of the patient.
Nurse and assistant positioning
The scrub nurse and the assistant surgeon stand on the opposite side with respect to the first surgeon.
Procedures
In the operating room she was monitored with 5 leads EKG, pulse oximetry and bispectral index. A large vascular access with a 16 Gouge intravenous cannula and invasive blood pressure has been positioned in the right radial artery.
With the patient in sitting position, the thoracic peridural was placed in the T6 space, placing the catheter no more than 5 cm from the entry point. The catheter suction test was negative for liquor aspiration and administration of 2 cc lidocaine 2% did not cause paresthesia in both patients.
A bolus of chirocaine 10 mg and sufentanil 10 mcg was administrated through the epidural catheter 20 minutes before the surgical incision.
One lung ventilation was started with 0.8 FiO2, 6 mL/kg tidal volume, PEEP 5 cmH2O and variable respiratory rate in order to keep arterial oxygen saturation above 90% and plateau pressure under 25 cmH2O.
Peridural top ups of chirocaine 7.5 mg and sufentanil 5 mcg were given when heart rate and systemic blood pressure increased 20% from baseline.
The continuous peridural infusion with 0.125% chirocaine and sufentanil 0.25 mcg/mL through elastomeric pump (6 mL/h) began at 2 hours from the surgical incision and continued for the following 72 hours.
The operations were performed using the uniportal video-assisted approach (9). After double tube ventilation a single 3–4 centimeter incision was made in the fifth intercostal space in between the mid and anterior axillary line.
Operation 1 (Figure 3)

A 30° angled-scope was used. The sequestration was located within the normal lower lobe without its own visceral pleura. The feeding arterial vessel was detected in the lower pulmonary ligament. After careful and gentle dissection with electro-cautery hook and ultrasonic scalpel, the artery was ligated proximally using a braided not-resorbable suture, then was cut with a vascular endostapler. Under gentle ventilation of the left lung we identified the non-ventilated area of the left lower lobe corresponding with the PS, and then a wide wedge resection was performed with endostapler. The surgical specimen was sent for pathological evaluation (Figure 1D). At the end of the procedure the arterial stump was covered with fibrin glue.
Operation 2 (Figure 4)

A 30° angled-scope was introduced in the chest. The ELS was clearly evident and demarcated from the normal lung tissue by a fissure of visceral pleura. Two independent feeding arteries, arising from descending aorta were identified within the lower pulmonary ligament. Feeding vessel dissection was performed using electric coagulation hook and ultrasonic dissector. The smaller feeding vessel was ligated and proximally and distally with braided not-resorbable sutures and then cut with scissors. The second bigger artery was ligated proximally with braided not-resorbable suture and then cut with a vascular endostapler. After that, the vein drainage, identified across the accessory fissure between the lower lobe and the ELS (Figure 2B), was dissected and stapled. The fissure between the PS and the left lower lobe was then divided with endostapler. The surgical specimen was removed and sent for pathological evaluation (Figure 2D).
A single 24-fr chest tube was used to drain the pleural space in both patients through the single incision. Chest drain suction at −20 cmH2O was maintained for 12 hours. At the end of the operation, the patient was extubated in the operation room and transferred to the hospital ward. Postoperative pain control was performed by injection of 1,000 mg acetaminophen every 8 hours, a rescue dose of NSAIDs (30 mg ketorolac) were administrated if visual analogue scale was >3. The chest tube was removed in second post-operative days as well the epidural catheter. The patients were discharged the day after chest tube removal in optimal clinical condition.
Discussion
PS is a congenital and histological benign clinical condition, but with potential life threatening complications. In literature, reported PS are complicated by massive hemoptysis, heart failure, systemic embolisms, sepsis, severe pulmonary infections etc. (4,5). In our case, the patient with ELS developed endocarditis with cerebral embolism. The patient with ILS manifested recurrent episodes of pulmonary infection and recurrent pneumonia. For those reasons, surgical resection of ILS and ELS is the preferred and definitive treatment of PS after the diagnosis is made. The preoperative work up of patients with PS requires precise study of the lung parenchyma with CT. Convectional CT scan doesn’t always demonstrate clearly the systemic artery, in particular in case in small vessels, therefore a careful evaluation of the abnormal vascular connection with angio-CT scan or angiography is required. Further effort should be made to exclude other congenital malformations that sometimes are associated with PS, such as congenital diaphragmatic hernia, vertebral anomalies, congenital heart disease, pulmonary hypoplasia and colon duplication (5). In our patients, pre-operative work up was completed with echocardiography, bronchoscopy, and total body CT-scan. Due to the experience acquired in the treatment of lung cancer we decided to perform surgery using the uniportal VATS approach despite in literature only a few case reports of uniportal VATS resection of PS were published (7,8). The first challenging step of the procedure was the isolation and closure of the systemic arterial branch. Following the previously reported technique of uniportal VATS (9,12), the movement of the instruments inside the chest were optimal and allowed us to identify ad isolate perfectly the vessels (13). In the first case, the anomalous blood supply comes from two arteries; in the second case, as usual, a single artery supplied the ILS. In our patients, the systemic arteries arise from the descending thoracic aorta. In both cases, the systemic arteries were ligated proximally with a braided non-resorbable suture. That gave us more security to avoid bleeding complications related to the fragility of these arterial vessels. The second step is the complete resection of the PS. In the case of ELS, the resection is made easier due to the clear demarcation by a fissure between the normal and affected tissue. In our ELS case, the uniportal approach allowed us an easier identification of the anatomical demarcation with the healthy lung and thus a complete resection of the ELS. Generally ILS is more technically demanding because of the lack of a clear anatomical demarcation with the normal lung and sometimes is deep in the normal lung tissue. Sometimes, a lobectomy is required to remove the ILS. Uniportal VATS has been demonstrated an effective approach to perform wedge resection and lobectomy for lung cancer (9,12,13). In our case, the ILS was peripheral and distant from the pulmonary hilum, and then it was removed with a wide wedge resection performed easily with endostapler through the uniportal approach.
In conclusion, we think that uniportal VATS is an effective and safe method to treat PS. This technique allows an ergonomic movement of the telescope and surgical instruments that doesn’t sacrifice the security of the procedure (9,13). Moreover, this is a benign condition generally manifested in young patients, thus uniportal VATS guarantees the better cosmetical results and less post-operative pain compared with the thoracotomy approach (Figure 5) (14).
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from both patients for publication of this report and any associated images and videos.
References
- Pryce DM. Lower accessory pulmonary artery with intralobar sequestration of the lung. A report of seven cases. J Pathol Bacteriol 1946;58:457-67. [Crossref] [PubMed]
- Markes C, Wiener SN, Reydman M. Pulmonary sequestration. Chest 1972;61:255-7. [PubMed]
- Albertini A, Dell'Amore A, Tripodi A, et al. Anomalous systemic arterial supply to the left lung base without sequestration. Heart Lung Circ 2008;17:505-7. [Crossref] [PubMed]
- Wei Y, Li F. Pulmonary sequestration: a retrospective analysis of 2625 cases in China. Eur J Cardiothorac Surg 2011;40:e39-42. [Crossref] [PubMed]
- Yucel O, Gurkok S, Gozubuyuk A, et al. Diagnosis and surgical treatment of pulmonary sequestration. Thorac Cardiovasc Surg 2008;56:154-7. [Crossref] [PubMed]
- Wan IY, Lee TW, Sihoe AD, et al. Video-assisted thoracic surgery lobectomy for pulmonary sequestration. Ann Thorac Surg 2002;73:639-40. [Crossref] [PubMed]
- Tamura M, Shimizu Y, Hashizume Y. Single-incision video-assisted thoracic resection for extrapulmonary sequestration: a case report. J Cardiothorac Surg 2014;9:22. [Crossref] [PubMed]
- Sihoe AD, Luo Q, Shao G, et al. Uniportal thoracoscopic lobectomy for intralobar pulmonary sequestration. J Cardiothorac Surg 2016;11:27. [Crossref] [PubMed]
- Gonzales-Rivas D, Paradela M, Fernandez R, et al. Uniportal video-assisted thoracoscopic lobectomy: two years of experience. Ann Thorac Surg 2013;95:426-32. [Crossref] [PubMed]
- Dell’Amore A, Giunta D, Campisi A, et al. Patient 1 Uniportal VATS resection of intralobar sequestration. Asvide 2018;5:382. Available online: http://www.asvide.com/article/view/23897
- Dell’Amore A, Giunta D, Campisi A, et al. Patient 2 Uniportal VATS resection of extralobar sequestration. Asvide 2018;5:383. Available online: http://www.asvide.com/article/view/23898
- Sihoe AD. The evolution of minimally invasive thoracic surgery: implications for the practice of uniportal thoracoscopic surgery. J Thorac Dis 2014;6:S604-17. [PubMed]
- Bertolaccini L, Rocco G, Viti A, et al. Geometrical characteristics of uniportal VATS. J Thorac Dis 2013;5 Suppl 3:S214-6. [PubMed]
- Bendixen M, Jørgensen OD, Kronborg C, et al. Postoperative pain and quality of life after lobectomy via video-assisted thoracoscopic surgery or anterolateral thoracotomy for early stage lung cancer: a randomised controlled trial. Lancet Oncol 2016;17:836-44. [Crossref] [PubMed]
Cite this article as: Dell’Amore A, Giunta D, Campisi A, Congiu S, Dolci G, Barbera NA, Agosti R, Buia F. Uniportal thoracoscopic resection of intralobar and extralobar pulmonary sequestration. J Vis Surg 2018;4:63.