Video-assisted thoracoscopic surgery lobectomy using “the caudal approach”: results and evolution
Introduction
In the last three decades, video-assisted thoracoscopic surgery (VATS) has become a common surgical approach in the diagnosis and treatment of lung and mediastinal diseases because of lower postoperative pain, less hospital stay and better cosmetic result (1-3). Nowadays, many case series have demonstrated the feasibility of VATS lobectomy since it was first reported by Roviaro in 1992 (4). In recent years, surgery has evolved towards the use of less thoracoscopic accesses driving the evolution of VATS from traditional three-port VATS (one utility incision of 4 cm and two ports of 1 cm) to biportal VATS (the utility incision and an additional thoracoscopic port), eventually to uniportal (only the utility incision). Despite biportal and uniportal VATS being approaches used all over the world (5-7), comparison between them and standard three-port VATS is not widely reported. We herein describe our current experience of thoracoscopic surgery using a new caudal position technique (8) and compared it with the standard anterior VATS technique (9).
Methods
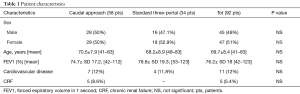
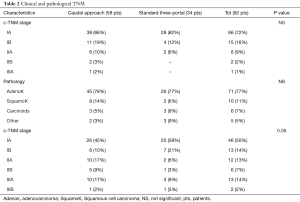
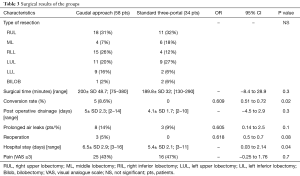
We retrospectively analyzed 92 consecutive patients who underwent VATS lobectomy between January 2016 and October 2017 in our institution. Among these, 58 patients were treated using the caudal approach to perform VATS lobectomy (8) and 34 were treated using the convectional Anterior three-port accesses to perform VATS lobectomy as described by Hansen et al. (9). Preoperative variables and intraoperative data were reported in Tables 1 and 2. For each patient, a retrospective data review including the following variables was made: gender, age at the time of surgery, preoperative FEV1, presence of cardiovascular comorbidity (defined as the presence of one or more of the following: congestive heart failure, ischemic cardiopathy with or without myocardial infarction, severe valvular cardiopathy, arrhythmia requiring chronic treatment, history of cerebrovascular disease, hypertension and/or peripheral vascular disease), presence of chronic renal failure, diabetes, clinical TNM stage (7th edition), pathology, type of surgical resection, pathological TNM staging (7th edition). Table 3 reported the post-operative variables used to compare caudal VATS lobectomy with standard anterior three-ports VATS lobectomy. The mean operative time, conversion rate, hospital stay, post-operative drainage, reoperation, post operative pain if ¡Ý3 using the visual analogue scale (VAS) reported the day of discharge and prolonged air leaks, defined as an air leaks for more than 3 days.
Full table
Full table
Full table
Operative technique
All patients underwent standard anesthesia care with the use of double lumen endotracheal tube. An epidural catheter for administration of analgesics was inserted in every patient.
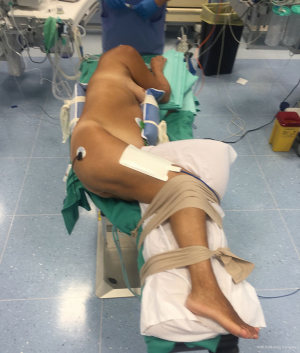
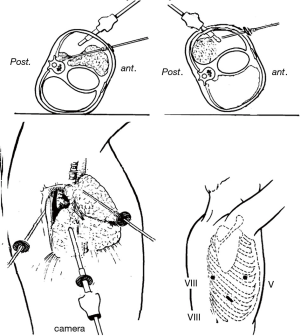
For caudal approach, the patient is placed in a lateral decubitus position with both legs flexed to the body by removing one of the two lower extremities of the operating bed (Figure 1); the first operator stays caudally the patient in the space created with the ability to have both hands equidistant from the ports. Our actual technique differs from the caudal approach we described before (8), indeed the incisions are made one of 1 cm at the 5th intercostal space (ICS), anterior axillary line, the second of 2–3 cm at the 8th ICS, middle axillary line and the third of 1 cm at the 8th ICS, posterior axillary line (Figure 2) (10), so the utility incision is made at the 8th ICS instead of the 4th one.

For standard three-port VATS, we follow the Copenhagen Approach (9) with a utility incision of 4 cm at the 4th ICS, between the breast and the lower angle of the scapula just anteriorly to the border of the latissimus dorsi muscle, and two thoracoscopic incision of 1 cm at the 7th or 8th ICS, anterior and posterior axillary lines. A wound protector was routinely used in both techniques.
All surgical instruments were exactly the same for both approaches. The operators used harmonic scalpel (Harmonic ACE®+, Ethicon Inc., Cincinnati, OH, USA) and thoracoscopic instruments for dissection and traction; the assistant controlled a 10-mm 30° thoracoscope and curved sucker to offer proper counter-traction and suction. Vascular and bronchial divisions were performed using mechanical endostapler (Endo GIA™ Ultra Universal Standard, Covidien Products—Medtronic; ECHELON FLEX™ ENDOPATH® Staplers, Ethicon—Johnson & Johnson Medical S.p.A) with sometimes curved-tip ones if difficult angles were encountered (Endo GIA™ curved tip reload with Tri-Staple™ technology, Covidien Products—Medtronic).
In all the patients we performed mediastinal lymph node dissection and, finally, a chest tube of 24–28 Fr was inserted through the inferior access.
Postoperative management
The patients were extubated in the operating room or on the same day in the intensive care unit. Postoperative pain relief was provided by continuous epidural administration of bupivacaine and intravenous paracetamol. Suction drainage at −20 cm H2O was used. For all the patients a digital chest drainage system was used (Thopaz™, Medela Italia Srl). Chest tube was removed when the pleural effusion was lower than 400 mL/day and air leak flow <40 mL/min for more than 8 h (and without spikes of airflow greater than this value).
Statistical analysis
All data for parametric continuous variables are reported as means ± standard deviation (SD). Significant differences between groups were assessed using Student’s t-test for continuous variables. Categorical variables are expressed as n (percentage) and were compared using χ2-test. Statistical analyses were performed using the SPSS software package version 20 (SPSS Inc., Chicago, IL, USA). A P value of <0.05 was considered significant.
Results
No statistical differences were reported between groups in terms of preoperative data and intraoperative entity of pulmonary resection as reported in Tables 1 and 2. Preoperative diagnosis was achieved in 33 patients (36%), 20 patients of caudal group (34%), 13 patients of anterior three-port group (38%). In 59 patients the diagnosis was achieved with fresh frozen section during surgery after pulmonary wedge resection. All patients had a neoplastic disease.
The patients’ perioperative outcomes are listed in Table 3. The surgical time, post-operative drainage, incidence of prolonged air leaks and post-operative pain were similar between groups, indeed these differences were not statistically significant. Reoperation was more frequent in the caudal group compared to the anterior three-port group, although this difference was not statistically significant (P=0.08). Reoperation was performed because of prolonged air leak (1 pt), post-operative bleeding (1 pt), torsion of the middle lobe (1 pt). Patients underwent caudal VATS lobectomy had a major risk for intraoperative conversion to open procedure (OR, 0.6; 95% CI, 0.51–0.72; P=0.02). The hospital stay was lower in the standard three-port group (5.4 vs. 6.5 days, P=0.04).We had no mortality.
Conclusions
These days, VATS-lobectomy has become a routine procedure for selected patients and has spread all over the world. However, there is no a standardized surgical technique. Many technical variations have been described in literature, showing differences in number of ports and their positions.
After we acquired good experience on VATS-lobectomy, we studied a new position in which the operator stays on the inferior side of the thorax, positioning the patient in a lateral sitting position.
We started to use the caudal position performing a three-port VATS and once we standardized the technique, we decided to compare this approach with a standard anterior three-port VATS-lobectomy. Our result showed caudal VATS to be feasible and equally safe to standard VATS, with no differences in operation time, chest drainage time, prolonged air leaks, postoperative pain.
Still considering similar disease and patient distribution, pathological TNM seems to be slightly different between the two groups but it's not statistically significant (P=0.06) and this parameter can partially justify the significant major risk of conversion to thoracotomy in the caudal group (P=0.02); moreover, all of them happened during the first tenth operations, so the difference between the two groups can be explained with the learning curve needed to improve this new approach.
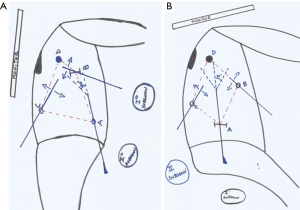
Considering the ergonomic characteristics of this new approach, we believe it can make it more comfortable for the surgeon. Ever since the first VATS operation, the strategy for port placement was unsettled. In the literature, it’s described as a placement that resembles a trapezoid shape (11), but it’s usually difficult to reproduce in the operating room resulting in several ergonomic problems (12). Surgeons often complain of fatigue of the arms and legs due to difficult and laborious movements of the upper limbs, wrists and shoulders. In fact, especially when reaching the posterior port, it’s necessary to rotate the torso, turn the neck and work in a different direction from the monitor and stay in an unstable position for quite some time (Figure 3A). As showed in the Figure 3B and Figure 4, our approach permits to stay in a straight position with almost all of the structures of the hilum and the monitor, with the upper limbs free to move perpendicularly to the field without complaining of excessive fatigue thanks to a more neutral position. Moreover, the neck and the head are always in straight position watching the monitor in front of the first surgeon over the patient’s head.
In conclusion, the caudal approach to major pulmonary resection results in a reliable, safe and comfortable procedure for the surgeon with intra- and perioperative results adhering to the various thoracoscopic approaches described in the literature. We use this approach as routine surgery and believe it’s simple and easy to reproduce. Once mastered on the caudal position with three ports, we started to perform lobectomies with a biportal and uniportal approach; in the first case, the incisions are made one of 2 cm at the 5th ICS, anterior axillary line, the second of 3 cm at the 8th ICS, middle axillary line (Figure 5) (13), in the second one, a single incision of 3 cm at the 8th ICS (Figure 6) (14). Anyhow, it is our intention to resubmit the above mentioned data after having studied a more numerous cohort of patients that have undergone such a surgery.


Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: No ethical committee approval and patient informed consent needed because of a retrospective study was conducted.
References
- Lewis RJ, Caccavale RJ, Sisler GE, et al. Video-assisted thoracic surgical resection of malignant lung tumors. J Thorac Cardiovasc Surg 1992;104:1679-85; discussion 1685-7.
- Palade E, Guenter J, Kirschbaum A, et al. Postoperative pain in the acute phase after surgery: VATS lobectomy vs. open lung resection - results of a prospective randomised trial. Zentralbl Chir 2014;139 Suppl 1:S59-66. [PubMed]
- Higuchi M, Yaginuma H, Yonechi A, et al. Long-term outcomes after video-assisted thoracic surgery (VATS) lobectomy versus lobectomy via open thoracotomy for clinical stage IA non-small cell lung cancer. J Cardiothorac Surg 2014;9:88. [Crossref] [PubMed]
- Roviaro G, Rebuffat C, Varoli F, et al. Videoendoscopic pulmonary lobectomy for cancer. Surg Laparosc Endosc 1992;2:244-7. [PubMed]
- Rocco G, Martucci N, La Manna C, et al. Ten-year experience on 644 patients undergoing single-port (uniportal) video-assisted thoracoscopic surgery. Ann Thorac Surg 2013;96:434-8. [Crossref] [PubMed]
- Gonzalez-Rivas D, Yang Y, Stupnik T, et al. Uniportal video-assisted thoracoscopic bronchovascular, tracheal and carinal sleeve resections†. Eur J Cardiothorac Surg 2016;49 Suppl 1:i6-16. [PubMed]
- Gonzalez D, Paradela M, Garcia J, et al. Single-port video-assisted thoracoscopic lobectomy. Interact Cardiovasc Thorac Surg 2011;12:514-5. [Crossref] [PubMed]
- Dolci G, Dell’Amore A, Daddi N. A new approach for video-assisted thoracoscopic lobectomy “the caudal position”. J Thorac Dis 2015;7:2348-51. [PubMed]
- Hansen HJ, Petersen RH, Christensen M. Video-assisted thoracoscopic surgery (VATS) lobectomy using a standardized anterior approach. Surg Endosc 2011;25:1263-9. [Crossref] [PubMed]
- Dolci G, Campisi A, Giunta D, Congiu S, Daddi N, Murana G, Dell’Amore A. VATS lobectomy using a three ports caudal approach. Asvide 2017;4:571. Available online: http://www.asvide.com/articles/1891
- Sihoe AD. The evolution of minimally invasive thoracic surgery: implications for the practice of uniportal thoracoscopic surgery. J Thorac Dis 2014;6:S604-17. [PubMed]
- Bertolaccini L, Viti A, Terzi A, et al. Geometric and ergonomic characteristics of the uniportal video-assisted thoracoscopic surgery (VATS) approach. Ann Cardiothorac Surg 2016;5:118-22. [Crossref] [PubMed]
- Dolci G, Campisi A, Giunta D, Congiu S, Daddi N, Murana G, Dell’Amore A. VATS lobectomy using a biportal caudal approach. Asvide 2017;4:572. Available online: http://www.asvide.com/articles/1892
- Dolci G, Campisi A, Giunta D, Congiu S, Daddi N, Murana G, Dell’Amore A. VATS lobectomy using a uniportal caudal approach. Asvide 2017;4:573. Available online: http://www.asvide.com/articles/1893
Cite this article as: Dolci G, Campisi A, Giunta D, Congiu S, Daddi N, Murana G, Dell’Amore A. Video-assisted thoracoscopic surgery lobectomy using “the caudal approach”: results and evolution. J Vis Surg 2017;3:187.