Bronchoscopic lung volume reduction with endobronchial valves for heterogeneous emphysema: long-term results
Introduction
Emphysema is a chronic obstructive pulmonary disease (COPD) characterized by irreparable damage of lung parenchyma with decrease in elastic recoil, progressive hyperinflation, and gas trapping. It represents the fourth most common cause of death according to the World Health Organization (WHO) (1). Treatment options include smoking cessation, inhaled medications, systemic corticosteroids, pulmonary rehabilitation, supplemental oxygen and lung transplantation. In selected cases, the resection of non-functioning areas of lung parenchyma, also named as lung volume reduction surgery (LVRS), has been reported to improve respiratory function. However, LVRS is a procedure associated with a significant morbidity (20–30%) and a relatively high mortality (7.9%) in the first 90 post-operative days (2). Thus, less invasive procedures are desirable considering the poor clinical condition of these patients. In the last years, bronchoscopic lung volume reduction (BLVR) with implantation of unidirectional EBVs has been proposed as a new treatment for emphysema. Clinical evidence from randomized controlled trials (RCTs) and retrospective case series showed that by achieving lobar occlusion in the absence of collateral ventilation (CV), lung volume reduction can be obtained with significant clinical responses in severe emphysematous patients (3-8). Despite these evidences, EBV is still being at the exploring stage. In the most of series the follow-up is limited to 12 months and the long-term efficacy of this procedure is poorly understood. Thus, in the present paper we aimed to evaluate the long-terms results and safety of BLVR with EBVs in consecutive patients with heterogeneous emphysema (HE).
Methods
Study design
This was a retrospective single center study performed at Thoracic Surgery Unit of University of Campania “Luigi Vanvitelli”, Naples, Italy. All consecutive patients with HE undergoing BLVR with EBVs (Zephyr TM EBV; Pulmonx Inc., Redwood, CA, USA) from January 2006 to December 2016 and completing at least 5 years of long-term follow-up were potentially eligible. Exclusion criteria were: (I) lack of complete clinical and functional follow-up data; (II) previous surgical procedures as LVRS, bullectomy and lung transplantation; (III) endoscopic hybrid procedures (i.e., EBV associated with insertion of coils or other devices). All patients were re-assessed from the functional point of view and quality of life (QoL) after 3, 6, 9, 12 months from the valve implantation, and then annually up to a maximum of 5 years. Radiologic monitoring was performed with Chest-X ray 24 hours after the procedure, at discharge, and 15 days later; and with high resolution computed tomography (HRCT) 1 and 3 months after BLVR and then on a yearly basis. Mortality, morbidity and survival were also recorded. The data were prospectively collected in a data-base and then retrospectively examined. The objectives were to evaluate the functional long-term results (primary endpoint) and the safe of the procedure (secondary endpoint). A written informed signed consent to receive EBV treatment was obtained by all patients who were also aware that their data could be used for scientific purpose only.
Study population
Forty-one patients undergoing EBV treatment for HE during the study period were potentially eligible. The selection criteria for EBV treatment were the same of endobronchial valve for emphysema palliation trial (VENT) study (6) as follows: (I) age of 40 to 75 years; (II) forced expiratory volume in one second (FEV1) <45% of predicted value; (III) total lung volume (TLV) >100% predicted; (IV) residual volume (RV) >150% predicted; (V) PaCO2 <50 mmHg (partial pressure of carbon dioxide in the blood); (VI) PaO2 >45 mmHg (partial pressure of oxygen in the blood); (VII) 6-minute walk test (6MWT) ¡Ý140 meters. In all cases, clinical examination, spirometry, PaO2 and PaCO2, diffusing lung capacity (DLCO); 6MWT; HRCT scan, QoL were evaluated before and after valve implant. In addition, morbidity and mortality were also recorded.
Pulmonary function test
Pulmonary function tests were attended according to American Thoracic Society (ATS) guidelines and included FEV1; forced vital capacity (FVC); RV; DLCO; 6MWT; PaO2 and PaCO2 measured at rest while breathing room air. All pulmonary function data are presented as a percentage of predicted values for the patient’s age, gender and height. Changes in patient’s QoL were measured by the St. George’s Respiratory Questionnaire (SGRQ), ranging from 0 to 100. The higher score indicated a worse clinical condition. All functional tests were attended before operation and 3, 6, 9, 12 months after the first valve implantation, and then annually up to a maximum of 5-year.
Radiological evaluation
All patients underwent a HRCT scan with volume rendering and three-dimensional reconstruction to define the more affected lobe to treat as previously reported (9). The percentage of heterogeneity was defined as the difference in the quantitative emphysema score (proportion of pixels of less than −910 Hounsfield units) between the target lobe and the adjacent parenchyma. Before treatment, the fissure integrity was measured on sagittal, axial, or coronal CT views and differentiated in fissure integrity ¡Ü75%; fissure integrity between 75–90%; and fissure integrity >90%. After EBV implant, the grade of atelectasis of the treated lobe was radiologically evaluated and patients were divided in “Collapse Group” if complete lobar atelectasis or moderate lobar collapse (collapse of more than 1 segment) were obtained and in “No-Collapse Group” if no atelectasis or collapse of only one segment were observed.
Device description
The last generation of Zephyr EBV valves was used in the presented study. It incorporated an unidirectional silicone valve that permitted to drain air and secretions from the distal lung segment during exhalation but prevented the entering of air during inhalation. During the study period, two sizes of valves were available in our hospital: a small valve (EBV 4.0) used for occluding bronchus having a diameter of 4.0–7.0 mm, and a large valve (EBV 5.5) for occluding bronchus with diameter of 5.5–8.5 mm.
Operative procedure
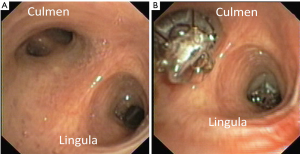
EBV insertion was applied with a flexible video-bronchoscopy (Olympus BF-1T160) under deep conscious sedation with spontaneous breathing, using short-acting benzodiazepine as midazolam and low dose propofol. General anaesthesia with endotracheal intubation was used only in particularly anxious patients to control the ventilation and prevent cough. After measuring the size of the bronchus with a dedicated catheter, the valve was loaded in the same catheter and then delivered in lobar, segmental, or sub-segmental bronchi, on the basis of individual anatomy, in order to completely occlude the target lobe. Antibiotics were administered before the procedure and in the first seven days after the procedure. An example is reported in Figure 1.
Statistical analysis
Data were summarized as mean and standard deviation (SD) for normally distributed continuous variables or absolute number and percentage for categorical variables. The survival was expressed as mean with 95% Confidence of Interval. The preoperative data were used as the baseline and compared with postoperative data by one-way ANOVA test corrected with Bonferroni post-hoc test, if indicated. Survival curves were calculated with Kaplan-Meyer and the difference evaluated with log-rank test. A p value 0.05 was considered significant. MedCalc statistical software (Version 12.3, Broekstraat 52; 9030 Mariakerke; Belgium) was used for the analysis.
Results
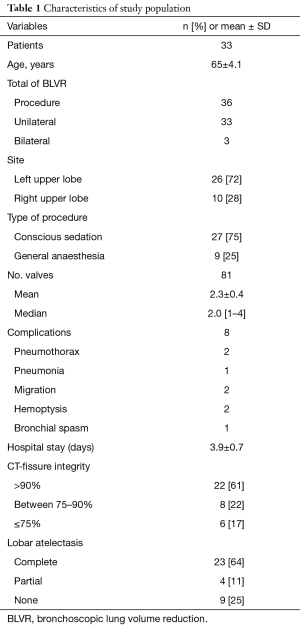
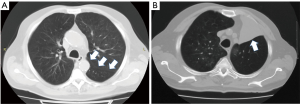
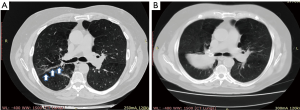
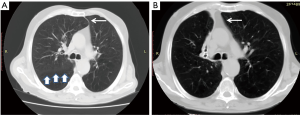
Of 41 eligible patients, 8 were excluded due to lack of data during follow-up (6 patients) or association with other surgical procedures (2 patients). Thus, our study population counted 33 patients, summarized in Table 1. Of these, 3/33 (9%) received a sequential contralateral BLVR procedure after a median interval of 23 months from the initial BLVR (range, 2–29 months) due to decline of respiratory function. These patients were also the object of a previous study (10). CT- fissure integrity >90% was found in 22 patients (61%); CT-fissure integrity between 75–90% was seen in 8 (22%) patients; and CT-fissure integrity ¡Ü75% was revealed in 6 (17%) patients. A total of 81 valves were deployed with a mean of 2.3±0.4 valves per patient. In 26 (72%) cases the left upper lobe and in 10 (28%) cases the right upper lobe were treated. The mean hospital stay was 3.9±0.7 days. Complete lobar atelectasis (Figure 2) was observed in 23 (64%) cases; partial lobar atelectasis in 4 (11%) cases (Figure 3); and no atelectasis in 9 (25%) cases (Figure 4). All patients of Collapse Group (n=27) presented a CT-fissure integrity >90% but one who had a CT-fissure integrity between 75–90%. Among 9 patients of No-Collapse Group, 6 (67%) presented a CT-fissure integrity ¡Ü75%; 2 (22%) a CT-fissure integrity between 75–90%; and 1 patient a CT-fissure integrity >90% (11%).
Full table
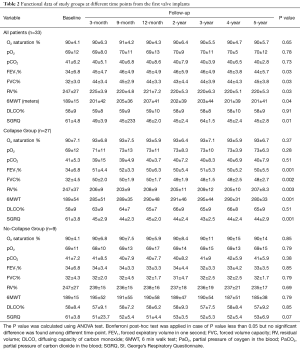
Functional assessment
Table 2 summarized the results. Among all patients, we observed a mild improvement of FEV1 (P=0.03); of FVC (P=0.03); of RV (P=0.03); of 6MWT (P=0.04) and SGQR (P=0.01). Collapse Group presented a significant improvement in FEV1% (+17%; P=0.001); in FVC (+18%; P=0.002); in RV (−39%; P=0.003); in 6MWT (P=0.001) and in SGRQ (P=0.001) after 12 months of follow-up. These results were retained for the entire follow-up without significant decline, as confirmed by Bonferroni post-hoc analysis. Conversely, EBV treatment did not show any significant benefits in No-Collapse Group.
Full table
Morbidity, mortality and survival
Morbidity
No major complications and death related to the procedure were observed. Two patients had pneumothorax treated with intercostal insertion of Heimlich valve; one patient had pneumonia that resolved with antibiotic therapy; two patients had hemoptysis that required the removal of the valves in one case; one patient had bronchial spasm that resolved with removal of valves three months later. In one patient undergoing bilateral BLVR procedure, a large valve migrated from the right upper lobe into the main left bronchus, three months after its insertion. It was removed and in the same setting three small valves (4.0 size) were replaced in the segments of right upper bronchus. In another patient, a small valve (4.0 size), placed within posterior segment of upper right bronchus, was expectorated two weeks later and then re-placed.
Survival
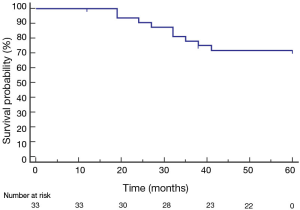
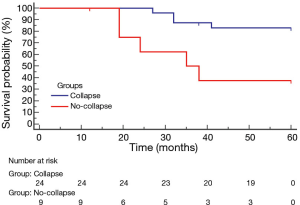
The mean survival was 51 months (95% CI: 46.5–56.3) (Figure 5). The 1-, 2-, 3-, 4- and 5-year survival rates were 100%, 90%, 78%, 71% and 71%, respectively. The death rate was 27% (9/33 patients). The cause of death was cancer (n=6); myocardial infarction (n=2) and end-stage respiratory failure (n=1). Patients with lobar collapse had a better survival than those without lobar collapse 55 months (95% CI: 51.3–59.5) versus 39 months (95% CI: 27.4–51.2); (P=0.001, hazard ratio: 4.2; 95% CI: 1.101–10.091, Figure 6).
Discussion
Emphysema is one of the leading causes of disability worldwide, despite the advancements in medical treatment and pulmonary rehabilitation. LVRS has been proposed as a feasible strategy to reduce the mismatch in size between the hyperinflated lungs and chest cavity and to restore the efficiency of respiration muscle activity. However, the significant morbidity and mortality related to LVRS have led physicians to explore less invasive procedures especially in high-risk surgical patients. Among these, BLVR with EBV is one of the most reported in literature (3-15) and its clinical benefits in selected patients is supported by five randomized studies, several cases series and meta-analysis (3-11). However, the most of series reported the short-term results of the EBV procedure (3-8) while the long-term results remained unexplored.
First, we found no significant positive effects in expiratory flow rates and 6MWT in No-Collapse Group while clinically important response with improvement in FEV1%, in RV%, in 6MWT, and in QoL was found in Collapse Group. The atelectasis of the more affected lobe simulated the physiological effect of LVRS proposed by Brantigan et al. (16). The lobar collapse obtained with valve implant diverted air to more perfused areas, recruited previously compressed alveoli, and facilitated the relatively healthier lung to function better. In addition, the reduction of over-inflated lung regions improved the diaphragm mechanic and the work of breathing. Our results are in line with previous studies. Unites States VENT study (3) and euro-VENT study (4) reported that patients with lobar exclusion and/or complete interlobar fissure had greater improvement in both FEV1 and 6MWT than counterparts. Wan et al. (17) found an improvement in FEV1 and 6MWT after EBV implant in all treated patients (n=96) but patients with complete lobar exclusion and value of FEV1% <30 vs. >30 had the greatest magnitude of benefit. The presence of complete inter-lobar fissure was the main predictive factor of lobar collapse. All patients but one with CT-fissure integrity >90% presented a complete collapse of target lobe. On the other hand, among 9 patients of No-Collapse Group, 6 had a CT-fissure integrity ¡Ü75%, two had a CT-fissure integrity between 75–90%, and one patient had a CT-fissure integrity >90%. In theory, the CV due to incomplete fissures precluded the lobar atelectasis of the treated lobe since air still entered through inter-lobar channels (Kohn pores) from adjacent non-treated lung. The inappropriate choice of valve size and/or the incorrect placement should be accepted as other reasonable explanations for the lack of lobar collapse especially in patient with complete fissure integrity. Our data are in line with the study of de Oliveira et al. (18) who retrospectively evaluated the prognostic value of CT-fissure integrity in 38/108 emphysematous patients undergoing EBV therapy. The Positive Predictive Values for lobar atelectasis were 83.9% for fissure integrity between 75–90%; 70% for fissure integrity ≤75%; and 90.5% for fissure integrity >90%. The Negative Predictive Value in case of fissure integrity <75% was 100%. Thus, in patients with incomplete fissure integrity the Chartis Pulmonary Assessment System could be indicated to measure CV before proceeding with EBV insertion. Herth et al. (19) reported that Chartis can predict the collapse of treated lobe after EBV implant with an accuracy of 75%. In BeLieVeR-HIFi study (7), post hoc analysis revealed that if the Chartis had been used to select the patients, the outcome would have been significantly better. Similarly, STELVIO (8), LIVE (5) and IMPACT (20) studies, where Chartis selected the patients to treat, showed that treated patients over control had a significant LVR and clinically meaningful benefits. Interestingly, in patients without lobar collapse a positive trend of subjective improvement in activity daily was seen, despite the absence of evident spirometric and radiological changes after procedure. Similarly, Wood et al. (21) found that some patients with clinical improvement after BLVR did not exhibit atelectasis or TLV reduction. Yet, Coxson et al. (22) found that significant improvements in QoL measured with SGRQ were not correlated with changes in standard function parameters. In theory, the low structural changes after procedure could help to redirect ventilation to better-perfused segments and improve ventilation-perfusion matching. In addition, the centralization of the mediastinum could be obtained also in absence of lobar atelectasis as shown in Figure 4 with improvement of respiratory mechanic.
Second, the early positive results were maintained during the entire follow-up in Collapse Group. Only three patients presented a decline of respiratory function that required a sequential contralateral treatment. This strategy allowed the recovery of the respiratory function at the same levels of the first procedure and prolonged the beneficial effects of BLVR. Also other authors (23,24) confirmed the long term effects of BLVR. Venuta et al. (23) in a cohort of 40 patients undergoing unilateral EBV implantation found significant improvements in spirometry, lung volumes, exercise capacity, breathlessness, and supplemental oxygen use at 5 years post insertion. Kotecha et al. (24) evaluated 16 patients undergoing EBV therapy with a follow-up more than 12 months and found that 6/16 (37.5%) patients had a long-term improvement in lung function.
Third, the procedure was safe also 5 years after valve implantation (25-27). The main complications were pneumothorax, haemoptysis, and valve dislocation. Pneumothorax is a well-known complication related to EBV implant due to change in the conformation of the lung after EBV insertion and the standard treatment consists in intercostal chest drainage. Haemoptysis is an expected adverse event following a thoracic procedure. Despite BLVR is a minimally invasive therapy, EBV may damage the bronchial wall and small vessels, inducing haemoptysis. It is often self-limiting but in some cases the removal of valves is needed as in one of our two cases. Valve migration has been previously reported without representing a major complication. Jenkins et al. (28) reported that two valves, inserted within lingular lobe and the anterior segment of the upper lobe for management of persistent air leaks after bullectomy, migrated to the orifice of the basal segment of the left and of the right lower lobe. In our series we observed two cases of valves migrations (29). In theory, the lack of elastic recoil due to severe progression of emphysema prevented any anchoring of valve to bronchial wall and facilitated migration. The mean survival was 51 months with a 1-,2-,3-,4- and 5-year survival rates of 100%, 90%, 78%, 71% and 71%, respectively. However, no case of death related to the procedure was observed. The United States VENT study (3) reported a rate of death of 2.6% at 6 months and 3.7% at 12 months and the European VENT study (4) showed a rate death of 5.4% at one year of follow-up. In the Stelvio study (8), the rate of death was 3% in 6 months and in BeLieVer HI-Fi study (7), it was 8%. We found that patients with lobar atelectasis presented a better survival compared to those without. Our results were also confirmed by other experiences. Hopkinson et al. (30) showed that all patients in whom radiological atelectasis had occurred (n=5) were alive 6 years after the procedure, whereas 8/14 patients without atelectasis had died raising the possibility that BLVR might, like LVRS, offer a survival advantage in appropriate selected patients. Venuta et al. (23) found that the survival rates after EBV insertion was higher in the groups of patients with complete fissures than those without on HRCT (83.3% vs. 24%).
In conclusion, our study confirms the general impression that BLVR with EBV implantation is viable strategy in management of HE and the early improvements of respiratory function could be maintained up to five years in patients with lobar collapse. The retrospective nature of the study, the small number of patients, the lack of Chartis measurement, the presence of patients undergoing sequential bilateral BLVR are all limitations that did not allow to draw definitive conclusions. Thus, our data should be confirmed by larger prospective study.
Acknowledgements
Alfonso Fiorelli thanks Francesca Del Regno for editing the paper.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study is a retrospective study. In addition we used for the treatment unidirectional Endobronchial valves that are used everywhere for the bronchoscopic management of emphysema. Thus, the approval from the institutional ethical committee is not required. A written informed signed consent to receive EBV treatment was obtained by all patients who were also aware that their data could be used for scientific purpose only.
References
- López-Campos JL, Tan W, Soriano JB. Global burden of COPD. Respirology 2016;21:14-23. [Crossref] [PubMed]
- Edwards MA, Hazelrigg S, Naunheim KS. The National Emphysema Treatment Trial: summary and update. Thorac Surg Clin 2009;19:169-85. [Crossref] [PubMed]
- Sciurba FC, Ernst A, Herth FJ, et al. A randomized study of endobronchial valves for advanced emphysema. N Engl J Med 2010;363:1233-44. [Crossref] [PubMed]
- Herth FJ, Noppen M, Valipour A, et al. Efficacy predictors of lung volume reduction with Zephyr valves in a European cohort. Eur Respir J 2012;39:1334-42. [Crossref] [PubMed]
- Skowasch D, Fertl A, Schwick B, et al. A Long-Term Follow-Up Investigation of Endobronchial Valves in Emphysema (the LIVE Study): Study Protocol and Six-Month Interim Analysis Results of a Prospective Five-Year Observational Study. Respiration 2016;92:118-26. [Crossref] [PubMed]
- Strange C, Herth FJ, Kovitz KL, et al. Design of the Endobronchial Valve for Emphysema Palliation Trial (VENT): a non-surgical method of lung volume reduction. BMC Pulm Med 2007;7:10. [Crossref] [PubMed]
- Davey C, Zoumot Z, Jordan S, et al. Bronchoscopic lung volume reduction with endobronchial valves for patients with heterogeneous emphysema and intact interlobar fissures (the BeLieVeR-HIFi study): a randomised controlled trial. Lancet 2015;386:1066-73. [Crossref] [PubMed]
- Klooster K, ten Hacken NH, Hartman JE, et al. Endobronchial Valves for Emphysema without Interlobar Collateral Ventilation. N Engl J Med 2015;373:2325-35. [Crossref] [PubMed]
- Fiorelli A, Petrillo M, Vicidomini G, et al. Quantitative assessment of emphysematous parenchyma using multidetector-row computed tomography in patients scheduled for endobronchial treatment with one-way valves†. Interact Cardiovasc Thorac Surg 2014;19:246-55. [Crossref] [PubMed]
- Fiorelli A, D'Andrilli A, Anile M, et al. Sequential Bilateral Bronchoscopic Lung Volume Reduction With One-Way Valves for Heterogeneous Emphysema. Ann Thorac Surg 2016;102:287-94. [Crossref] [PubMed]
- Liu H, Xu M, Xie Y, et al. Efficacy and safety of endobronchial valves for advanced emphysema: a meta analysis. J Thorac Dis 2015;7:320-8. [PubMed]
- Santini M, Fiorello A, Di Crescenzo VG, et al. Use of unidirectional endobronchial valves for the treatment of giant emphysematous bulla. J Thorac Cardiovasc Surg 2010;139:224-6. [Crossref] [PubMed]
- Santini M, Fiorelli A, Vicidomini G, et al. Endobronchial treatment of giant emphysematous bullae with one-way valves: a new approach for surgically unfit patients. Eur J Cardiothorac Surg 2011;40:1425-31. [Crossref] [PubMed]
- Santini M, Fiorelli A, Vicidomini G, et al. Iatrogenic air leak successfully treated by bronchoscopic placement of unidirectional endobronchial valves. Ann Thorac Surg 2010;89:2007-10. [Crossref] [PubMed]
- Fiorelli A, Costanzo S, Carelli E, et al. Bronchoscopic treatment of complex persistent air leaks with endobronchial one-way valves. Gen Thorac Cardiovasc Surg 2016;64:234-8. [Crossref] [PubMed]
- Brantigan OC, Mueller E. Surgical treatment of pulmonary emphysema. Am Surg 1957;23:789-804. [PubMed]
- Wan IY, Toma TP, Geddes DM, et al. Bronchoscopic lung volume reduction for end-stage emphysema: report on the first 98 patients. Chest 2006;129:518-26. [Crossref] [PubMed]
- de Oliveira HG, de Oliveira SM, Rambo RR, et al. Fissure Integrity and Volume Reduction in Emphysema: A Retrospective Study. Respiration 2016;91:471-9. [Crossref] [PubMed]
- Herth FJ, Eberhardt R, Gompelmann D, et al. Radiological and clinical outcomes of using Chartis™ to plan endobronchial valve treatment. Eur Respir J 2013;41:302-8. [Crossref] [PubMed]
- Valipour A, Slebos DJ, Herth F, et al. Endobronchial Valve Therapy in Patients with Homogeneous Emphysema. Results from the IMPACT Study. Am J Respir Crit Care Med 2016;194:1073-82. [Crossref] [PubMed]
- Wood DE, McKenna RJ Jr, Yusen RD, et al. A multicenter trial of an intrabronchial valve for treatment of severe emphysema. J Thorac Cardiovasc Surg 2007;133:65-73. [Crossref] [PubMed]
- Coxson HO, Whittall KP, Nakano Y, et al. Selection of patients for lung volume reduction surgery using a power law analysis of the computed tomographic scan. Thorax 2003;58:510-4. [Crossref] [PubMed]
- Venuta F, Anile M, Diso D, et al. Long-term follow-up after bronchoscopic lung volume reduction in patients with emphysema. Eur Respir J 2012;39:1084-9. [Crossref] [PubMed]
- Kotecha S, Westall GP, Holsworth L, et al. Long-term outcomes from bronchoscopic lung volume reduction using a bronchial prosthesis. Respirology 2011;16:167-73. [Crossref] [PubMed]
- Fiorelli A, Costanzo S, di Costanzo E, et al. The early detection of lung cancer during follow-up of patients undergoing endobronchial one-way valve treatment for emphysema. Arch Bronconeumol 2015;51:e13-5. [PubMed]
- Fiorelli A, Accardo M, Vicidomini G, et al. LigaSure meets endobronchial valve in a case of lung cancer with pneumoconiosis. Transl Lung Cancer Res 2013;2:308-10. [PubMed]
- Fiorelli A, Santini M. Bronchoscopic lung volume reduction: an alternative to repeated lung volume reduction surgery. Eur J Cardiothorac Surg 2016;50:584. [Crossref] [PubMed]
- Jenkins M, Vaughan P, Place D, et al. Endobronchial valve migration. Eur J Cardiothorac Surg 2011;40:1258-60. [PubMed]
- Fiorelli A, Reginelli A, Santini M. Endobronchial Valve Migration: A "Whistle Blower". J Bronchology Interv Pulmonol 2016;23:e24-6. [Crossref] [PubMed]
- Hopkinson NS, Kemp SV, Toma TP, et al. Atelectasis and survival after bronchoscopic lung volume reduction for COPD. Eur Respir J 2011;37:1346-51. [Crossref] [PubMed]
Cite this article as: Fiorelli A, Santoriello C, De Felice A, Ferrigno F, Carlucci A, De Ruberto E, Mastromarino R, Occhiati L, Messina G, Santoriello E, Vicidomini G, Polverino M, Santini M. Bronchoscopic lung volume reduction with endobronchial valves for heterogeneous emphysema: long-term results. J Vis Surg 2017;3:170.