Unusual case of subxiphoid uniportal VATS right upper lobectomy in a patient with interrupted inferior vena cava with azygous continuation
Case presentation
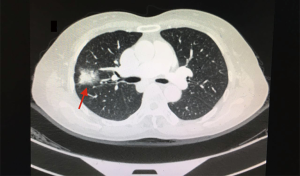
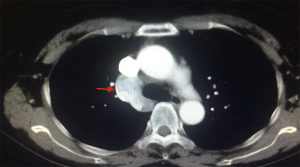
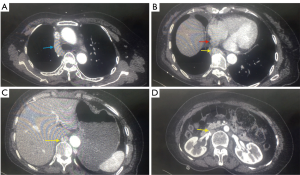
A 66 year-old non-smoker female patient, presented to our hospital with a history of cough for the last month. The patient had unremarkable Physical examination. Laboratory tests showed no abnormalities. ECG showed normal pattern. Chest radiograph revealed a solid (3 cm °¡ 3.5 cm) lesion in the right upper lobe the lung with slight widening of the mediastinum and enlargement of the azygos arch, CT scan confirmed the findings (Figure 1) and PET-CT showed pathological uptake in the lesion while the mediastinum was free. The arch of the azygos vein was abnormally dilated (Figure 2) and the IVC was shown in the right hemithorax posterior to the left atrium of the heart and lateral to the esophagus (Figure 3). The hepatic veins joined and drained to the cardiac right atrium in a direct fashion (Figure 4). The patient was scheduled to undergo a surgery of right upper lobectomy through the subxiphoid uniportal VATS approach.
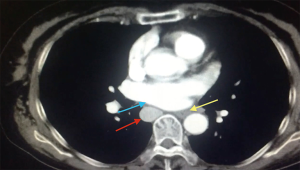
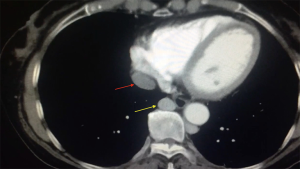
Preoperative preparation
Other abdominal abnormalities were excluded by abdominal CT scan. Echocardiography revealed no cardiac abnormality, the operation was scheduled to be performed via a minimal invasive technique.
Equipment performance card
Full HD thoracoscopic camera (Karl Storz®), wound protectors, special instruments designed for VATS surgery (Shanghai Medical Instruments Group Ltd.), Endo GIA staplers (Covidien-Medtronic), LigaSure™ energy device (Covidien-Medtronic).
Procedure
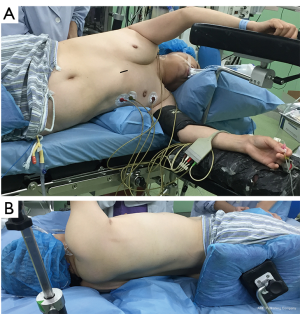
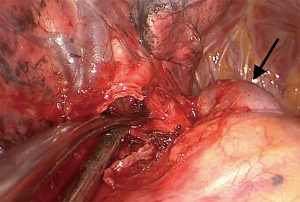
Under general anesthesia, the patient was intubated with a double lumen endotracheal tube (32 Fr). CVP line, arterial line, and urinary catheter were inserted before beginning of the surgery. The patient was positioned on left decubitus position, with slight backward tilting (70°) (Figure 5). A 3.5 cm longitudinal skin incision were performed over the xiphoid prominence, the xiphoid process was resected thereafter using surgical scissors. The right pleura were opened, and the pericardial fats were removed in order to improve the exposure for the pleural space (Figure 6). The right hilum was approached by dividing the parietal pleura around the right upper lobe venous branches. This was done by special long instruments with a curved tip which was specially designed for subxiphoid surgeries (Figure 7). The venous branch to the middle lobe was identified, and the truncus anterior branch of the pulmonary artery was exposed. The vein and the artery were encircled and divided using a white vascular stapler with the angled tip (Endo GIA staplers Covidien-Medtronic) (Figure 6). The abnormally dilated azygos arch was noticed during the procedure (Figure 8) and dissections near to the azygos arch where done carefully in order to avoid its injury. The bronchus to the upper lobe was subsequently dissected and encircled, inflation test where done to insure the patency of bronchus intermedius and the bronchus was stapled using a green reload stapler (Endo GIA staplers Covidien-Medtronic) (Figure 6). The posterior ascending artery was identified, dissected, encircled and tied using double 2-0 silk ties then the artery was divided by LigaSure™ energy device. The interlobar fissure then divided using two green reload staplers (Endo GIA staplers Covidien-Medtronic), and the specimen was removed out of the pleural cavity. Posterior retraction of the IVC with a sucker was necessary in order to perform lymph node dissection at station 8 and station 7 (Figure 9). Lymph nodes at station 3a were easily dissected and removed (Figure 6). For dissecting the lymph nodes at station R4 and R2 the azygos arch was encircled and retracted laterally using a metal wire that was percutaneously inserted (Figure 10), then lymphadenectomy was done. After hemostasis and pneumostasis one chest drain was inserted through the same subxiphoid incision, and the wound was closed in layers.

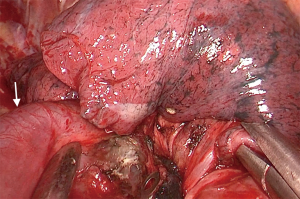
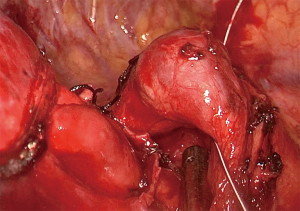
Post-operative management
Postoperative course was uneventful. Chest drain was removed in the third postoperative day, and the patient was discharged from the hospital four days after the operation. Follow up CT was performed one month after the surgery and revealed no other abnormalities (Figure 11). The patient was in a good general condition and asymptomatic in a clinic visit.
Discussion
In the field of thoracic surgery, pulmonary and systemic anatomical vascular variations are not a rare condition. Interrupted IVC (also known as azygos continuation of the IVC is a relatively uncommon congenital condition with prevalence 1.5% (0.2–3%) of the general population (2-4). During the sixth to tenth weeks of gestational life, three paired venous systems develop to form the retroperitoneal venous system: posterior cardinal (which arise first at the sixth week), followed by the subcardinal, and supracardinal systems at seven weeks, and eight weeks. The IVC pre-renal segment originated from the subcardinal system. While the renal vein segment originates as a result of subcardinal-supracardinal systems anastomosis. The azygos and hemiazygos veins arise from the supra-cardinals just above the diaphragm. Changes and modulations occur in the abdominal and pelvic basic venous system resulting in variants and anomalies such as isolated left IVC, double IVC, and retro aortic left renal vein (5). In azygos continuation of the IVC, the pre-renal IVC passes posterior to the diaphragmatic crura to enter the thorax as the azygos vein (2). The embryologic theory suggests atrophy of the right subcardinal vein due to subcardinal-hepatic anastomosis failure, which is responsible for the atrophy of right subcardinal vein. As a result, the supra-subcardinal anastomosis shunts the blood through the retro crural azygos vein (2,6). The renal portion of the IVC receives blood return from the kidneys and passes cephalad to the thorax as the azygos vein. The azygos vein drains to the SVC at the usual anatomical point in the right paratracheal area. In fact the hepatic segment is not truly absent; rather, it drains directly into the right atrium of the heart instead of the IVC (2). The IVC is usually interrupted after the entry of the renal veins and continues into a dilated azygos vein that enters the right pleural space, runs posteriorly in the mediastinum and joins to the superior vena cava in the right paratracheal area (7,8). In rare cases, the continuation of the IVC could occur at the left side through a hemiazygos venous system. The hepatic veins usually drains to the post- hepatic segment of the IVC which is almost invariably present (8). This anomaly has been incidentally reported for the first time in 1793 at post-mortem examination (9). In 1967, Moller and his group (10), described the relationship between interrupted IVC and the polysplenia syndrome after studying the anatomical data of twelve cases of concomitant visceral heterotaxy with polysplenia. Subsequently, several reports have confirmed this pertinence (11-13). Although some cardiac anomalies such as the polysplenia syndrome, A-V septal defects, anomalous of pulmonary venous connection and pulmonary atresia could be presents in combination with interrupted IVC but it’s not common to this anomaly to produce physiological consequences (8). In the past, predominant relationship between the anomaly and congenital heart disease thought to be exist (8), but with the development of cross-sectional imaging, more cases has become recognized as a solitary finding in otherwise asymptomatic patients (7). P wave aberration and multiple atrial arrhythmias are frequently seen in the ECG of these patients (14). Recognizing the dilated azygos vein at the confluence with the superior vena cava and in the retro crural space is important to avoid misdiagnosis as a paratracheal mass or retro crural lymph nodes enlargement (6,7). Technical attention is required during cardiac surgeries performed for other conditions and during right sided thoracic procedures, and awareness of this anomaly is paramount in order to avoid damage or ligating the azygos vein which would be invariably lethal (3,4). Video-assisted thoracic surgery (VATS) has become a popular procedure in thoracic surgery. Although VATS provides to surgeons the ability to obtain better details of local structures while performing the operation, the lack of wide view of the surgical field during the operation considered as one of its limitations (15,16). It is important to acquire preoperative accurate information on the vascular anatomy as it may serve to avert such complications and boost the safety of the surgery. To our knowledge, there are no video documented reports of this abnormality. Moreover this is probably the first case with such abnormality approached by VATS.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Abu Akar FE, Yang C, Zhou Y, et al. Subxiphoid RUL lobectomy in a patient with interrupted IVC with azygos continuation. Asvide 2017;4:283. Available online: http://www.asvide.com/articles/1592
- Bass JE, Redwine MD, Kramer LA, et al. Spectrum of congenital anomalies of the inferior vena cava: cross-sectional imaging findings. Radiographics 2000;20:639-52. [Crossref] [PubMed]
- Hardwick T, Belcher E, Sabharwal T, et al. Interrupted inferior vena cava: high-risk anatomy for right thoracotomy. Interact Cardiovasc Thorac Surg 2011;12:850-2. [Crossref] [PubMed]
- Effler DB, Greer AE, Sifers EC. Anomaly of the vena cava inferior; report of fatality after ligation. J Am Med Assoc 1951;146:1321-2. [Crossref] [PubMed]
- Malaki M, Willis AP, Jones RG. Congenital anomalies of the inferior vena cava. Clin Radiol 2012;67:165-71. [Crossref] [PubMed]
- Ginaldi S, Chuang VP, Wallace S. Absence of hepatic segment of the inferior vena cava with azygous continuation. J Comput Assist Tomogr 1980;4:112-4. [Crossref] [PubMed]
- Schultz CL, Morrison S, Bryan PJ. Azygos continuation of the inferior vena cava: demonstration by NMR imaging. J Comput Assist Tomogr 1984;8:774-6. [Crossref] [PubMed]
- Mazzucco A, Bortolotti U, Stellin G, et al. Anomalies of the systemic venous return: a review. J Card Surg 1990;5:122-33. [Crossref] [PubMed]
- Abernethy J, Banks J. Account of Two Instances of Uncommon Formation, in the Viscera of the Human Body. By Mr. John Abernethy, Assistant Surgeon to St. Bartholomew's Hospital. Communicated by Sir Joseph Banks, Bart. P. R. S. Philosophical Transactions of the Royal Society of London 1793;83:59-66. [Crossref]
- Moller JH, Nakib A, Anderson RC, et al. Congenital cardiac disease associated with polysplenia. A developmental complex of bilateral "left-sidedness". Circulation 1967;36:789-99. [Crossref] [PubMed]
- Rose V, Izukawa T, Moës CA. Syndromes of asplenia and polysplenia. A review of cardiac and non-cardiac malformations in 60 cases withspecial reference to diagnosis and prognosis. Br Heart J 1975;37:840-52. [Crossref] [PubMed]
- Van Mierop LH, Gessner IH, Schiebler GL. Asplenia and polysplenia syndromes. Birth Defects 1972;8:36-44.
- Van Praagh S, Santini F, Sanders SP. Cardiac malpositions with special emphasis on visceral heterotaxy (asplenia and polysplenia syndromes). In: Fyler DC. editor. Nadas’ Pediatric Cardiology. Philadelphia: Hanley and Belfus, 1992:589-608.
- Van der Horst RL, Gotsman MS. Abnormalities of atrial depolarization in infradiaphragmatic interruption of inferior vena cava. Br Heart J 1972;34:295-300. [Crossref] [PubMed]
- Matsumoto K, Yamasaki N, Tsuchiya T, et al. Three-dimensional computed tomography for a mediastinal basal pulmonary artery. Ann Thorac Surg 2012;94:e115-6. [Crossref] [PubMed]
- Watanabe S, Arai K, Watanabe T, et al. Use of three-dimensional computed tomographic angiography of pulmonary vessels for lung resections. Ann Thorac Surg 2003;75:388-92; discussion 392. [Crossref] [PubMed]
Cite this article as: Abu Akar FE, Yang C, Zhou Y, Lin L, Gonzalez-Rivas D, Jiang L. Unusual case of subxiphoid uniportal VATS right upper lobectomy in a patient with interrupted inferior vena cava with azygous continuation. J Vis Surg 2017;3:92.