Robotic thymectomy—a new approach for thymus
Introduction
The gold standard technique for thymectomy has been transsternal approach. The main advantages of this technique are asserted to have an optimal exposure and availability of the complete dissection of the thymus and mediastinal fatty tissue (1). By this way, the risk of possible incomplete thymectomy has been claimed to be zero, a complete healing from myasthenia gravis is possible (1). The risks of major vascular and phrenic nerve injuries are very low (1). Major disadvantages of this technique include that: splitting of the sternum, the longer duration of operation and postoperative hospitalization. For these reasons transsternal resections for nonthymomatous thymus have almost been tailed in major thoracic surgery centers.
One of the most commonly used approaches is the transcervical thymectomy. It is a minimally invasive technique which is mostly preferred by younger females and neurologists (2). The advantages of transcervical thymectomy are short hospitalization, and fewer complications (2). However, the main criticism includes the incomplete resection of thymus or perithymic fatty tissue due to crowding of instruments.
Video-assisted thoracic surgery (VATS) thymectomy gained popularity after 2000s. It can be performed via the left- or right-sided approach or even subxiphoidal or bilateral (3). The disadvantages of this technique are the 2-dimensional view of the operative field and the long learning curve (3).
Recently, robotic-assisted thoracic surgery (RATS) has become into as an alternative approach to either, open surgery or video-assisted thoracoscopic surgery. Resection of thymus in the treatment of myasthenia gravis and thymoma is also a new era for robotic-assisted surgery. A detailed radiologic examination is essential for selection of appropriate case for surgery also to decide the surgical approach. Patients with Myasthenia Gravis are examined by a qualified neurologist and anesthesiologist before surgery.
Our robotic (da Vinci Systems Intuitive Surgical, Sunnyvale, California) thymectomy technique with tricks is explained in detail in this presentation.
Preparation of surgery—positioning of the patient and docking of the robot
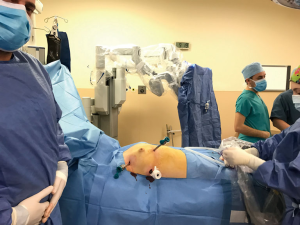
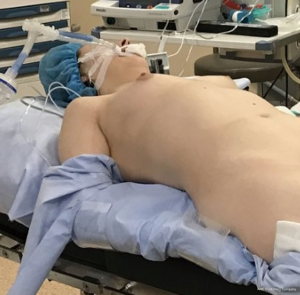
The right sided approach is preferred due to our long lasting experience with VATS. We give the patient a 30-degree semi-supine position. The patient is supported with a roll placed under the right shoulder, and the right arm is draped and positioned inferiorly near the chest (Figure 1).
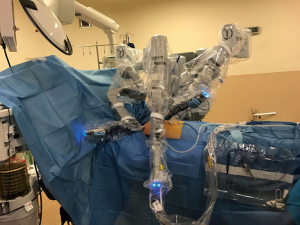
A double lumen endotracheal tube is placed with the help of a bronchoscope. Three ports are used in this surgery. The incisions are performed around the breast without violating the mammalian tissue (Figure 2). The first port is always the camera port and zero degree camera is used while placing the other ports. During the operation 30 degrees camera should be used when necessary. The left port is opened in the anterior axillary fossa and the right port is opened in the 5–6th intercostal space under the breast folds. We do not open an access port unless the operation is performed for a thymoma resection. On thymoma resections the left port is enlarged and an Alexis retractor (Applied Medical, Rancho Santa Margarita, CA, USA) is replaced. After the placement of the ports, side docking of the robot is performed (Figure 3).
Surgical technique
We use carbon dioxide insufflation with a pressure of 6 mmHg until we open contralateral mediastinal pleura. In the left arm a prograsper is used and for the right arm Maryland forceps is preferred. After the careful exploration of the cavity and the phrenic nerve, we begin the dissection with resection of the right sided pericardiophrenic fatty tissue. The resection of the thymus begins by dissecting the thymus on the pericardium anterior to the phrenic nerve with blunt dissection or Maryland forceps in the right hand (Figure 4).

Then the thymus is dissected from the sternum by opening the contralateral mediastinal pleura. The dissection on the superior vena cava parallel to the phrenic nerve enables visualization of the superior vena cava and junction of both innominate veins. The dissection of the upper poles needs caution. By gentle traction on the superior poles caudally, both superior poles of the thymus could be dissected separately from their attachments to thyrothymic ligament with their capsule without violating of the thymic tissue (Figure 5). The arterial and venous branches could be identified and divided during this maneuver. The major thymic veins may be at different numbers and locations and caution is needed to divide them carefully without causing hemorrhage (Figure 6).


After complete dissection of the upper poles, the thymus is retracted caudally, as follows it became completely freed from left innominate vein. The left side of the thymus could be dissected from the pericardium by pulling the thymic tissue toward the surgeon. By this way, the left phrenic nerve could be visualized. After completion of the left thymus resection, fatty tissue located at the left pericardiophrenic angle is completely resected.
The specimen is removed with an Endo bag (Covidien, USA) from the axillary port if an access port was not opened. The mediastinum is carefully inspected for any remaining fatty mediastinal tissue and hemostasis. One 10 mm Jackson Pratt drain is placed through the most anterior port across the mediastinum to drain the both chest.
Tips and tricks in resection of thymomas
If the operation is performed for a thymoma resection, the left port is selected as access port, because intercostal space is larger in this area and assistance could be performed from this port by an Alexis retractor. The thymoma should be resected at the last part of the operation and a non-touch technique should be performed during the whole surgery in robotic thymothymectomies, similar with VATS thymoma operations. For this reason, the non tumourous part of the thymus is dissected first and these tissues are used for grasping and traction.
Discussion
Despite the median sternotomy has been the gold standard for a long time (1,7), in the past 20 years, minimal invasive approaches have become accepted techniques for thymectomy (8).
In patients with thymoma the International Thymic Malignancy Interest Group (ITMIG) recommends a complete thymectomy for patients without myasthenia gravis (MG) and extended thymectomy for patients with MG. This kind of resections either prevent possible recurrences, or increase the possible remission rates (9). A neurological benefit and decreased use of steroids in the majority of patients after thymectomy has been reported (10). Incomplete thymectomy operations are unacceptable in patients with MG. This includes either the patient has a thymoma or not (11).
Robot technology is an evolution of manual videothoracoscopy introduced to overcome limitations of videothoracoscopic surgery such as rigid instruments and suboptimal vision (12). More intuitive movements, tremor filtration, more degrees of manipulative freedom, motion scaling, and high-definition stereoscopic vision are advantages of the robotic approach (13). As a consequence of these superiorities of robotic surgery, performing safer and complete thymus resections can be feasible.
Robotic thymectomy is a feasible and safe surgical technique with comparable perioperative outcomes to the open surgery in patients. A learning curve of 15–20 cases may be required by the surgeons to safely perform this relatively novel technique (14). Robotic thymectomy with good perioperative outcomes obtained, especially the team is experienced in videothoracoscopic surgery.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Jaretzki A 3rd, Wolff M. "Maximal" thymectomy for myasthenia gravis. Surgical anatomy and operative technique. J Thorac Cardiovasc Surg 1988;96:711-6. [PubMed]
- Calhoun RF, Ritter JH, Guthrie TJ, et al. Results of transcervical thymectomy for myasthenia gravis in 100 consecutive patients. Ann Surg 1999;230:555-9;discussion 559-61. [Crossref] [PubMed]
- Mineo TC, Pompeo E, Lerut TE, et al. Thoracoscopic thymectomy in autoimmune myasthesia: results of left-sided approach. Ann Thorac Surg 2000;69:1537-41. [Crossref] [PubMed]
- Kaba E, Cosgun T, Ayalp K, et al. Maryland bipolar forceps of da Vinci is used to dissect out the gland from the pericardium and sternum without disturbing the integrity of the capsule. Asvide 2017;4:197. Available online: http://www.asvide.com/articles/1506
- Kaba E, Cosgun T, Ayalp K, et al. Respectively right upper pole then left upper pole are retracted by using a moderate amount of tension. Asvide 2017;4:198. Available online: http://www.asvide.com/articles/1508
- Kaba E, Cosgun T, Ayalp K, et al. The major thymic vein is preparing. Asvide 2017;4:199. Available online: http://www.asvide.com/articles/1509
- Daniel VC, Wright CD. Extended transsternal thymectomy. Thorac Surg Clin 2010;20:245-52. [Crossref] [PubMed]
- Suda T, Sugimura H, Tochii D, et al. Single-port thymectomy through an infrasternal approach. Ann Thorac Surg 2012;93:334-6. [Crossref] [PubMed]
- Bae MK, Lee SK, Kim HY, et al. Recurrence after thymoma resection according to the extent of the resection. J Cardiothorac Surg 2014;9:51. [Crossref] [PubMed]
- Keijzers M, de Baets M, Hochstenbag M, et al. Robotic thymectomy in patients with myasthenia gravis: neurological and surgical outcomes. Eur J Cardiothorac Surg 2015;48:40-5. [Crossref] [PubMed]
- Friedant AJ, Handorf EA, Su S, et al. Minimally Invasive versus Open Thymectomy for Thymic Malignancies: Systematic Review and Meta-Analysis. J Thorac Oncol 2016;11:30-8. [Crossref] [PubMed]
- Veronesi G. Robotic lobectomy and segmentectomy for lung cancer: results and operating technique. J Thorac Dis 2015;7:S122-30. [PubMed]
- Veronesi G, Cerfolio R, Cingolani R, et al. Report on First International Workshop on Robotic Surgery in Thoracic Oncology. Front Oncol 2016;6:214. [Crossref] [PubMed]
- Kamel MK, Rahouma M, Stiles BM, et al. Robotic Thymectomy: Learning Curve and Associated Perioperative Outcomes. J Laparoendosc Adv Surg Tech A 2017. [Epub ahead of print]. [Crossref] [PubMed]
Cite this article as: Kaba E, Cosgun T, Ayalp K, Alomari MR, Toker A. Robotic thymectomy—a new approach for thymus. J Vis Surg 2017;3:67.