Non-intubated uniportal left-lower lobe upper segmentectomy (S6)
Introduction
Although considered inferior to lobectomy in the treatment of primary lung cancer during many years, anatomical segmentectomies have been recently included as an alternative treatment in specific subtypes of primary lung adenocarcinomas (1). Secondarily, more interest has been focused in sublobar resections for many indications such as benign conditions, pulmonary metastasis and those subtypes of early stage primary adenocarcinoma (2-6). Although initially considered challenging, non-intubated anatomical segmentectomies have gained popularity in the last 5 years, mainly in Asia where anatomical patient features and a different pattern in non-small cell lung cancer (NSCLC) have allowed an evolution in non-intubated sublobar resections (7,8). Here we report a non-intubated uniportal VATS left-lower lobe (LLL) upper segmentectomy after chemotherapy within a two-stage treatment of bilateral pulmonary metastasis.
Case presentation
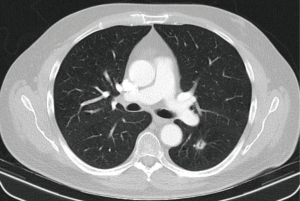
A 62-year-old man with history of arterial hypertension, diabetes mellitus and previous smoker, was treated for a sigma adenocarcinoma in 2011 with surgery, and received adjuvant chemotherapy. After 24 months of follow-up, bilateral pulmonary nodules and mediastinal lymph nodes appeared, and after a first negative biopsy, in 2014 a new fine-needle biopsy evidenced pulmonary metastasis, so chemotherapy plus antiangiogenic treatment were administered, and with evidence of stable disease and two PET positive residual pulmonary nodules in right-upper lobe (RUL) and LLL (Figure 1), surgery of both lesions was indicated.
As the RUL was initially accessible for a wedge resection, LLL nodules was approached first. The patient was not obese (BMI 25), had normal FEV1 and DLCO values, and without anatomical limitations, we suggested a non-intubated uniportal VATS S6 LLL anatomical segmentectomy in April 2016. Deep sedation was achieved with remifentanil infusion 0.05–0.1 μg/kg/min and intravenous bolus of 1–2 mg of midazolam. Routine monitoring and BIS TM Quatro bispectral index between 40 and 60 was routinely applied to monitor the level of consciousness. During the operation, the patient was placed in the right lateral decubitus position; high-flow oxygen nasal prongs at 40 liter per minute (lpm), and non-invasive ETCO2 and invasive radial artery monitoring to control oxygen saturation and avoid excessive hypercapnia. A quiet surrounding atmosphere was created and material for a potential emergent situation was carefully set up. A 4 cm incision in the 5th intercostal space was performed and then covered by a wound protector retractor. An iatrogenic pneumothorax was created after dissecting into the chest cavity and the lung collapsed gradually while the patient remained spontaneously breathing. Intercostal blockade with Bupivacaine 0.5% 1–1.5 cc/space from the 3rd to the 7th space (Figure 2) and vagal block in the aortopulmonary window with 3–4 cc Bupivacaine 0.5% were done as routinely (Figure 3). A left-upper lobe wedge resection of a subcentimetric nodule was performed first, with intraoperative pathologic result without evidence of malignancy (bronchiectasis). Then, anatomical S6 segmentectomy was completed, and we did not perform systematic lymph node dissection (Figure 4). Air leak was checked by insufflating the lung through a facial mask under saline solution. Total operation time was 240 minutes (induction plus recovery time, and surgical time 25 and 215 minutes respectively), lowest oxygen saturation was 97%, and highest pCO2 was 54 mmHg. Respiratory and hemodynamic parameters during the operation are reflected in Table 1.




Full table
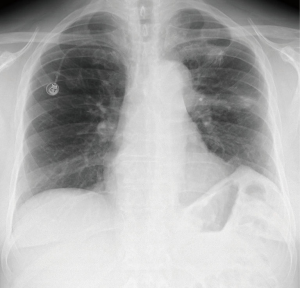
Patient resumed oral intake 6 hours after the surgery, and began walking at postoperative day 1 (POD 1). The was no air leak so chest tube was removed at 24 hours, and patient was discharged home on POD 2 with normal chest X-ray (Figure 5). Pain measured with visual analogue scale (VAS) was 2, 3 and 2 at 6, 24 and 48 hours respectively.
Final pathologic exam was concordant with intestinal metastasis of 1.5 cm. And surgical margins of 5 mm. The patient is doing his normal life and continues follow-up with medical oncology.
Conclusions
Sublobar anatomical segmentectomies are nowadays indicated for early stage primary adenocarcinomas, as well as pulmonary metastasis and benign conditions (12). These lung-sparing resections are especially useful in pulmonary metastasis as they preserve healthier parenchyma and functional reserve, avoiding a pulmonary lobectomy in cases where central lesions contraindicate a wedge resection (13). Experience and a comprehensive segmental anatomical knowledge are mandatory in order to avoid complications. These procedures have spread specially in Asia, where predominance of small adenocarcinomas in a non-smoker non-obese population has fed the interest of thoracic surgeons in less aggressive lung-preserving surgery.
Performing the procedure in a spontaneously breathing patient has the potential advantage of faster resumption of oral intake and mobilization and a decrease of postoperative morbidity, and also shortens the chest drain length and hospital stay (7,14), thus it has been included as an enhanced recovery after surgery (ERAS) pathway for improving patient care. On the other hand, there is very little evidence about the oncological outcomes regarding on the hypothetical minor alteration of the antitumor immune response. Indications of non-intubated lung-preserving resections are continuously growing as the most innovative surgical teams develop more and more procedures under safety conditions with excellent results (15).
Non-obese patients without pulmonary underlying disease, where pulmonary metastasis develop after or during primary site treatment, seem a good patient profile for non-intubated operations regarding the better anatomical and functional status. Preoperative chemotherapy within a systemic multimodal treatment does not represent a limitation and shouldn’t preclude the surgery as it does not limit the procedure technically.
Uniportal VATS has proved feasible and safe for almost every pulmonary resection, including non-intubated sublobar resections (16), thus becoming an ideal approach in expert hands in order to decrease the aggressiveness of global patient surgical care.
Investigation beyond this point focuses on acquiring evidence of quality about its comparative benefits over standard intubated procedures.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- NCCN Guidelines version 3.2017. Non-small cell lung cancer. National Comprehensive Cancer Network. Available online: https://www.nccn.org
- Speicher PJ, Gu L, Gulack BC, et al. Sublobar Resection for Clinical Stage IA Non-small-cell Lung Cancer in the United States. Clin Lung Cancer 2016;17:47-55. [Crossref] [PubMed]
- Khullar OV, Liu Y, Gillespie T, et al. Survival After Sublobar Resection versus Lobectomy for Clinical Stage IA Lung Cancer: An Analysis from the National Cancer Data Base. J Thorac Oncol 2015;10:1625-33. [Crossref] [PubMed]
- Cao C, Chandrakumar D, Gupta S, et al. Could less be more?-A systematic review and meta-analysis of sublobar resections versus lobectomy for non-small cell lung cancer according to patient selection. Lung Cancer 2015;89:121-32. [Crossref] [PubMed]
- Macke RA, Schuchert MJ, Odell DD, et al. Parenchymal preserving anatomic resections result in less pulmonary function loss in patients with Stage I non-small cell lung cancer. J Cardiothorac Surg 2015;10:49. [Crossref] [PubMed]
- Sesti J, Donington JS. Sublobar Resection: Ongoing Controversy for Treatment for Stage I Non-Small Cell Lung Cancer. Thorac Surg Clin 2016;26:251-9. [Crossref] [PubMed]
- Guo Z, Yin W, Pan H, et al. Video-assisted thoracoscopic surgery segmentectomy by non-intubated or intubated anesthesia: a comparative analysis of short-term outcome. J Thorac Dis 2016;8:359-68. [Crossref] [PubMed]
- Liu J, Cui F, He J. Non-intubated video-assisted thoracoscopic surgery anatomical resections: a new perspective for treatment of lung cancer. Ann Transl Med 2015;3:102. [PubMed]
- Galvez C, Navarro-Martinez J, Bolufer S, et al. Video-assisted intercostal blockades with Bupivacaine. Asvide 2017;4:134. Available online: http://www.asvide.com/articles/1442
- Galvez C, Navarro-Martinez J, Bolufer S, et al. Video-assisted vagal blockade with Bupivacaine. Asvide 2017;4:135. Available online: http://www.asvide.com/articles/1443
- Galvez C, Navarro-Martinez J, Bolufer S, et al. Anatomical upper segmentectomy of the left lower lobe through uniportal VATS in a spontaneously breathing patient. Asvide 2017;4:136. Available online: http://www.asvide.com/articles/1444
- McDonald F, De Waele M, Hendriks LE, et al. Management of stage I and II nonsmall cell lung cancer. Eur Respir J 2017;49:1600764. [Crossref] [PubMed]
- Yoshimoto K, Nomori H, Mori T, et al. Postoperative change in pulmonary function of the ipsilateral preserved lung after segmentectomy versus lobectomy. Eur J Cardiothorac Surg 2010;37:36-9. [Crossref] [PubMed]
- Liu J, Cui F, Li S, et al. Nonintubated video-assisted thoracoscopic surgery under epidural anesthesia compared with conventional anesthetic option: a randomized control study. Surg Innov 2015;22:123-30. [Crossref] [PubMed]
- Peng G, Cui F, Ang KL, et al. Non-intubated combined with video-assisted thoracoscopic in carinal reconstruction. J Thorac Dis 2016;8:586-93. [Crossref] [PubMed]
- Hung MH, Cheng YJ, Hsu HH, et al. Nonintubated uniportal thoracoscopic segmentectomy for lung cancer. J Thorac Cardiovasc Surg 2014;148:e234-5. [Crossref] [PubMed]
Cite this article as: Galvez C, Navarro-Martinez J, Bolufer S, Sesma J, Lirio F, Galiana M, Rivera MJ. Non-intubated uniportal left-lower lobe upper segmentectomy (S6). J Vis Surg 2017;3:48.