From full thoracotomy to uniportal video-assisted thoracic surgery: lessons learned
Introduction
Changing in surgery is a must. Reducing scars and the insult of surgical trauma has become a vital end point of the entire surgical assessment and endeavour. In the pursuit of further reduction in scarring and the trauma of both mini-thoracotomy and conventional VATS, surgeons and Industry have combined their ingeniousness and expertise to promote two new approaches for VATS: uniportal VATS (uniVATS) and awake thoracic surgery procedures (1). UniVATS allows performing the same procedure as three-port VATS through only one incision, usually along the anterior axillary line. The most recent development of non-intubated anesthetic techniques applied to uniVATS could enhance the concept of minimal invasiveness (1). Nowadays, the combination of uniVATS and awake surgery is perceived by thoracic surgeons as the final stage of minimally invasive thoracic surgery, a stage leading to fast-tracking in thoracic surgery. This stage should be not so difficult to reach if we think that this approach originates from a simple advancement of the anterior multiport VATS approach, the expansion of the uniVATS for procedures characterized by minor or intermediate difficulty (2), but, also as a direct evolution from anterior limited thoracotomies.
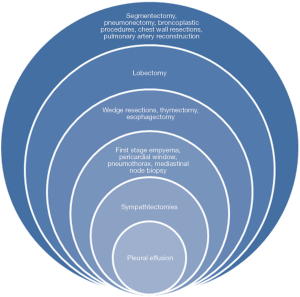
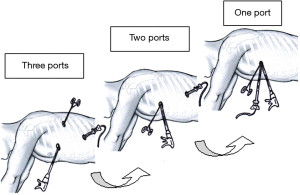
While posterior VATS approaches never really became widely performed even after more than 20 years of being described, the anterior approach alternative has finally been adopted in recent years as a valid widely offered alternative to thoracotomy (3) (Figure 1).The advantages of VATS lung resections compared to current minithoracotomy from a functional standpoint are becoming very obvious, albeit relatively short-lived (4). In the future, we are likely to read publications assessing prospectively survival in well-designed comparative studies any differences comparing the two approaches. In this setting, the VIOLET multicenter study from the UK will contribute to assess the superiority of VATS lobectomy over thoracotomy (5). At present, uniportal robotic surgery is still under investigation as a real added value to minimally invasive surgical thoracic approaches. This article will discuss our experience in developing uniVATS from full thoracotomy and three-port VATS with details regarding lessons learned from transition in a surgical approach less and less invasive (Figure 2).
Patient selection, workup and preoperative preparation
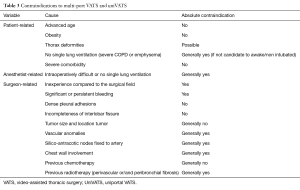
Minimally invasive thoracic surgery represents a strategic choice at the institutional level. Along with the consideration for local socioeconomic issues (6), the choice of VATS over thoracotomy should be based on a series of important criteria (Table 1).

Full table
- Clinical criteria:
- Obesity (evaluation of BMI): obtaining access for lung and mediastinal dissection can be difficult in patients with significant subcutaneous obesity planned for a VATS or a uniVATS. For these patients, longer trocars (up to 10 cm) can provide easier access into the chest. A wound protector may also adequately retract subcutaneous tissue in the access incision. Patients with significant abdominal obesity may require more superior port placement or a moderately reversed Trendelenburg position to lessen the effect of cranially displaced diaphragms; access for left thoracoscopic surgery may be compromised by the smaller operative thoracic space. Full lung deflation should occur prior to dissection, and contralateral tidal volumes should be minimized. Rolling the operating table can also allow gravity to improve lung retraction;
- Limited pulmonary function (7): VATS is useful patients with limited pulmonary function have better outcomes when surgery is performed via VATS compared with traditional open techniques; high risk patients could take advantage, more than fit patients, of minimally invasive surgery.
- Radiological criteria:
- Characteristics of pulmonary lesions: volume of tumor, hilar position, across complete/incomplete fissures;
- FDG-PET: standard uptake value which may direct the need for evaluation of the mediastinal nodal compartment prior to VATS (see below);
- 3D reconstruction (8) has been proven to be useful in order for thoracic surgeons to perform appropriate anatomical segmentectomy and curative resections. The software allows extractions of the lung field region, pulmonary artery, pulmonary vein, bronchus, and other regions using contrasted CT images, and the result is displayed in 3D maps.
- Endoscopic criteria:
- Bronchoscopic anatomy. Preoperative bronchoscopy is mandatory: bronchial anomalies and unexpected endobronchial lesions may be a factor in the decision-making process;
- Nodal evaluation through TBNA-EBUS: to better select patients with early stage NSCLC for surgery and to reveal the presence of possible antracotic nodes which increase the risk of conversion.
- Intraoperative findings:
- Patient safety and oncological adequacy. Is patient safety put at risk? It is recommendable to be aware that conversion to thoracotomy at the correct time during the learning curve indicates prudence not failure. In addition to being easier on patients, minimally invasive techniques should not compromise the safety or quality of the cancer operation when applied appropriately;
- Adhesions. Dense adhesions (previous open chest surgery, prior radiation to the chest, history of pulmonary infections, prior coronary artery bypass graft (CABG) surgery may affect the choice for VATS especially in the steep part of the learning curve;
- Intraoperative nodule localization. In the absence of a preoperative diagnosis and if the lesion cannot be readily found, several techniques have been developed to facilitate intra-operative localization of SPN during VATS (9). Methylene blue injection carries the risk of spreading the colorant on the pleural surface or difficulty to identify the lesion in patients with extensive anthracotic pigments. Specialized equipment, such as CT-fluoroscopy (10) or gamma probe, is required in case of injection of specific radiotracer. Intra-operative ultrasound detection requires a specific flexible ultrasound probe and is operator dependent (11). In non-collapsed lung or emphysematous patients the localization of SPN is also limited. For nodules not amenable to finger palpation (<10 mm nodules located at the distance of more than 5 mm from the pleural surface or >10 mm deeply located nodules situated at a distance of >10 mm from the visceral pleura), VATS resection after CT-guided hook wire localization for SPN remains the most diffuse method to localize a pulmonary nodule (9).
- Oncological criteria:
- Surgical margins. Are surgical clear margins guaranteed in patients with lung cancer? Newly published reports are confirming a similar rate of completely excised procedures with clear margins between uniVATS and other approaches (12);
- Nodal staging. Nowadays, there is little doubt that adequate lymphadenectomy can be performed via VATS surgery. In most reports comparing uniportal and multiportal VATS surgeons have demonstrated at least as extensive mediastinal exploration as with any approach (13).
- Surgeon experience:
- Surgical and staff skills. Different studies support the need to have a dedicated surgical and anesthesiological team (3); furthermore, within a surgical team, different individuals may evolve to different degrees according to individual skills and talents. One third of patients could be considered to be relatively complex case for an initial program of VATS because of: strong pleural adhesion, silico-anthracotic adherent lymph nodes, incomplete or fused fissures, pneumoconiosis, tumor size (larger than ≥5 cm), BMI >30, vascular abnormalities of the pulmonary artery branches (14).
Instrumentation and equipment preference card for uniVATS
UniVATS means simultaneous introduction of instruments parallel to the videothoracoscope through a single port. Handling the videothoracoscope by the assistant surgeon is aimed at visualizing the position of the instruments at any time during the procedure with simple movements. This modality of surgical approach seems to offer an advantage to surgeons with its familiar field of direct view as in open approach. The uniportal approach enables surgeon to overcome the problem, characteristically related to three port VATS, of a dihedral angle that represents an obstacle to the depth visualization. This geometric configuration (15) renders uniVATS similar to open surgery and allow surgeon to cranially suspend a target parenchimal area so to have a lateral as well as back-to-front displacement to easily perform wedge resections. As far as the instruments are concerned, they could be similar to those used in conventional VATS. The uniportal equipment preference card has addressed technology towards instruments reserved to simplify and improve outcomes of this surgical procedure (Table 2).
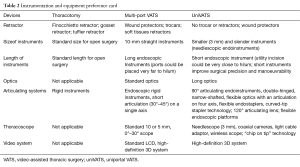
Full table
Theoretically, triangulation in standard VATS permits traction on tissues to facilitate dissection along normal anatomical planes. One of the main problems that single-port surgery brought along with lack of triangulation was “swordfighting” or the “chopsticks” effect as the instruments going in close to each other clashed with one another and the camera head and telescope. Articulating instrumentation allows for pseudotriangulation to occur intracorporeally despite the entry points being adjacent to one another through the same skin incision. A few novel ideas like pre-bent/articulated instruments have been developed with this type of surgery (16). Articulating instruments (in particular endostaplers and endograspers), endoscissors and needle holders are available. The additional development of the “curved tip” staple end for vascular endoGIA, is a further feature that allows the staple to pass through narrower angles. Articulating instruments have a 0–90 ° range of motion, allowing infinite freedom for tip adjustment, thanks also to ability to rotate their intrathoracic parts of 360°. They have a spin lock mechanism that allows them to be used it as a rigid instrument. The disadvantage is that using all these articulating instruments has a significant learning curve before one handles them dexterously. All these difficulties may result in crossing of instruments at times (cross-triangulation), a frequently used manoeuvre in single-incision surgery. As one gets familiarised with this surgery, it is easy to see that an instrument passed from the left end of the port automatically gravitates to the right of the intrathoracic target organ and vice versa. For the intrathoracic visualization, a 5-mm, 0° or 30° videothoracoscopes represent, in our opinion, the best option, with a preference for the latter. The problem of the perpendicular light cable of regular thoracoscopes and the bulky camera heads clashing with the instruments can be done away with by the use of a telescope with a coaxial light cable or simply a longer scope so that the camera head moves away from the surgeon’s hands. The Endo Eye (Olympus, Tokyo, Japan) was suited for this as it came with the “chip-on-tip” technology which meant that it had a streamlined profile with a single coaxial cable and this reduced the cluttering and clashing with the bulky camera head (16). It also came with a deflectable tip. The use of this type of thoracoscope does have a learning curve for the assistant holding the camera. Alternately, surgeon could choose to hold the scope and, in the same time, to do the dissection with the other hand. Furthermore, the versatility and range of view given by these new generation scopes could minimize chest trauma by reducing the need for torqueing at the wound.
Technical aspects
Single port surgery dates back to the very beginning of thoracic surgery. In our previous Institution of affiliation dedicated to the cure of tuberculosis and its sequelae, single port thoracoscopy (also called pleuroscopy) had been performed since the late 80’s (17). Our idea of a single port in thoracic surgery has been the natural translation of a large previous surgical experience in medical thoracoscopy and in vats sympathectomies.
In 2004 Rocco et al. described and then popularized the geometric rationale of a novel technique to perform pulmonary wedge resections through a single thoracic incision, with the assistance of a 5 millimeter thoracoscope and the use of articulated instrumentation (2). The theoretical and practical aspects of the so-called uni-portal VATS were better explained in another paper published in 2005 (18) by the same group of authors. The advantage of uniVATS, is that the vision is directed to the target area, bringing the instruments to address the target lesion from a straight perspective and obtaining similar angle of view as for open surgery. As reported by the senior author (19), in the uniVATS the surgeon and assistant tend to be positioned together in front of the patient that lies in a lateral position with a hyperextension of the chest wall. The single port technique relies on a sort of fulcrum of the operative instruments inside the chest and lets the surgeon work along the sagittal/caudo-cranial plane he/she is mostly accustomed to in open. The main screen is placed directly in front of the operating surgeon supporting the impression of the “direct” approach of uniVATS. As the incision is placed at the anterior axillary line, the position of the camera (the surgeon’s eye) lies very close to the dome of the thoracic cavity when the patient is placed in the lateral position, and at mid-point between the apex and the base of the cavity allowing close access to all areas. The placement of the incision is of paramount importance to guarantee the success of the procedure. The majority of target lesions in the chest can be addressed by selecting intercostal space incision between the fourth and the sixth, in the midaxillary line. The general principle to consider is “if you can see your target, you can also reach it.” Therefore, the first rule is an adequate position of the thoracoscope. In a football stadium analogy, the camera plan is structured so to have an adequate view on the whole field. The privileged visual point is undoubtedly represented by the central sector (VIP sector). From here, the football players will be ever under our control; in the same way, all peripheral nodules in the lung can be theoretically resected by a uniportal approach (20).
The “sine qua non” conditions for developing of uniVATS are similar to those which have facilitated multi-port VATS worldwide spreading:
- Safety: patient safety should be the first and foremost cornerstone of making progress on a minimally-invasive procedure; VATS pulmonary resections for lung cancer has less morbidity compared to open approaches (21); avoiding an open approach may improve outcomes, although conversion for safety or complete resection can be necessary;
- Feasibility: uniVATS can be performed by surgeons experienced with three or double-port technique and with documented experience in open surgery both anterior (22) and posterolateral thoracotomy; there are evidences that it is possible to perform a direct transition from a full thoracotomy to the single port with as satisfactory postoperative results as with the transition from multiport technique (23);
- Reproducibility: several series of uniVATS lung resections confirms the reproducibility of this technique (24); surgeons are at ease with this method even without a 3-port VATS experience (23);
- Efficacy: demonstration by VATS surgeons that there are at least equal achievements to open surgery in obtaining clear margins, exploring and retrieving of hilar and mediastinal nodes, number of postoperative complications (25).
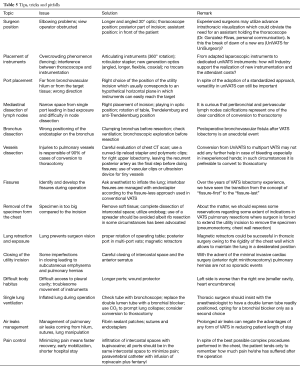
Several maneuvers acquired from experience with both open and three-port VATS lobectomy can reduce the technical difficulty encountered in uniVATS and often prevent conversion to an open approach. Nevertheless, real life surgery teaches us that there are some barriers that prevent an endoscopic procedure (Table 3).
Full table
Mini-thoracotomy and uniVATS: the same perspective changing philosophy
Nowadays, uniVATS means one single incision on the chest, generally positioned anteriorly, smaller than 5 cm, usually from IV to V intercostals space. The entity of incision should be dependent on the kind of procedure: uniVATS for minor/intermediate procedure should require an incision of not more than 2–3 cm, whereas uniVATS for major pulmonary resection should require an incision as not more than 5 cm. In addition, single port incision should be performed with the intent to avoid nerve injury (electrocautery, rib spreading with instruments, retractors) and to obtain a total muscle-sparing (moving muscle without cutting the fibers).
Therefore, we reiterate that uniVATS has a well-established surgical identity not to be confused with a “muscle-sparing” minithoracotomy.
Several thoracic units around the world (23) have easily experienced a direct transition from thoracotomy/minithoracotomy to uniVATS, without previous experience in VATS procedures. This observation further reiterates the idea that uniVATS is not so different to an open approach and the transition to uniVATS could be less traumatic than to a multi-port VATS.
Most of published reports compare outcomes between multi-port VATS and open thoracotomy. Obviously, comparing VATS with any open approach, most important outcomes (pain, chest drain, hospital long stay, morbidity and mortality) are in favour of the minimally invasive approach. Thinking about the homogeneity of two surgical techniques to compare, minithoracotomy and uniVATS stand out as the perfect setting. UniVATS is totally similar to an open approach, except for the entity of incision and the lack of rib retraction. View perspective and instrumentation angles (sagittal plane) are similar to open procedures when performed through an antero-lateral thoracotomy/minithoracotomy.
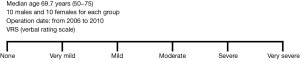
Our experience with both minithoracotomy and uniVATS, the latter already reported in the setting of already published by current author (26), led to obtain preliminary information in terms of main outcomes (postoperative pain and costs);these data were used for an internal institutional audit. We retrospectively reviewed patients who underwent atypical wedge pulmonary resections for stage I NSCLC between January 2006 and December 2010, at the National Cancer Institute, IRCCS Pascale Foundation of Naples. During that period, we identified 40 consecutive patients who underwent uniVATS wedge resection (20 patients) and Minithoracotomy wedge resection (20 patients) and survived at least one year. We performed a case-matched analysis comparing the two groups based on a series of preoperative variables. There were no differences in patient sex (10 males and 10 females for each group), age (median age 69.7 years (range, 50–75 years), comorbidities (Charlson score) and preoperative pulmonary function. Patients have been evaluated for early (<30 days) and late (after one year) postoperative pain measured by the verbal rating scale (VRS) (Figure 3). Results are reported in Figure 4 and Table 4. The difference in perceived pain became more significant as more time elapsed from the intervention date. In our opinion, this analysis has many limitations (i.e., its retrospective nature and small numerosity) among which the most important being the lack of information about postoperative paresthesia (27). However, the difference of long –term residual postoperative wound pain is so striking that warrants additional investigation. In this same context, we also compared the minithoracotomy to uniVATS wedge resections for lung cancer in terms of costs based on the total number of procedures performed in our Institution. We estimated all itemized costs related to each procedure and observed a significant difference in costs favoring uniVATS wedge resections mainly due to the reduced hospitalization compared to the open technique (Table 4).
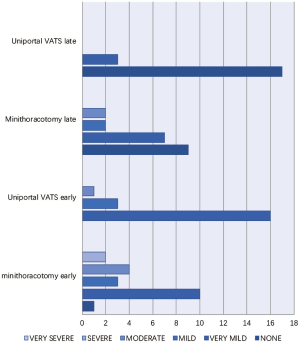
Full table
Postoperative management
If conventional multiportal VATS are superior to open thoracotomy by virtue of minimizing surgical access trauma (28), then further reduction in uniVATS should yield even greater benefits. Therefore, reducing the number of wounds from three or four to just one, should, in theory, lead to a less pain (29), paresthesia (27), morbidity (27) and faster recovery (30). Both short and long term pain following surgery have been proven to be improved when VATS is performed instead of open surgery. The vast majority of these reports involve multiport VATS (31). More recently, some degree of reduced postoperative pain after single port VATS in comparison to multiport VATS has been described (32).
Perna et al. (33) recently have published a prospective randomized study comparing uniVATS with other styles of VATS. They employed a sound process of randomization, a comprehensive selection of outcome measures and a thorough analysis of the collected data. From this, they have reached a conclusion that uniVATS offers no measurable benefits compared with other VATS approaches when performing lung lobectomy. The limited number of patients, the arbitrary patient selection putting the Duke technique (essentially a variant of uniportal technique) with the Copenhagen technique vs. uniVATS, the conclusion of “non-superiority” of uniVATS compared to other VATS in terms of different postoperative outcomes (postoperative pain, time of removing paravertebral catheter and chest drain, duration of postoperative hospital stay, postoperative complications and mortality), are limitations that urgently require more studies from more centers in order to validate these results.
In this light, the meta-analysis authored by Harris et al. (29) aimed to compare the clinical outcomes of uniVATS and multiportal VATS lobectomy for patients with lung cancer. Interestingly, all eight studies included for the systematic review were from Eastern countries. This meta-analysis demonstrated favorable outcomes for uniVATS lobectomy in the treatment of lung cancer compared to the conventional multiport approach.
Tips, tricks and pitfalls
In a troubleshooting table (Table 5), we offer ‘tips’ to both avoid and manage numerous intra-operative technical difficulties that commonly arise during multi-port VATS and uniVATS.
Full table
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Gonzalez-Rivas D, Bonome C, Fieira E, et al. Non-intubated video-assisted thoracoscopic lung resections: the future of thoracic surgery? Eur J Cardiothorac Surg 2016;49:721-31. [Crossref] [PubMed]
- Rocco G, Martin-Ucar A, Passera E. Uniportal VATS wedge pulmonary resections. Ann Thorac Surg 2004;77:726-8. [Crossref] [PubMed]
- Petersen RH, Hansen HJ. Learning thoracoscopic lobectomy. Eur J Cardiothorac Surg 2010;37:516-20. [Crossref] [PubMed]
- Whitson BA, Groth SS, Duval SJ, et al. Surgery for early-stage non-small cell lung cancer: a systematic review of the video-assisted thoracoscopic surgery versus thoracotomy approaches to lobectomy. Ann Thorac Surg 2008;86:2008-16; discussion 2016-8.
- Lim E, Paramasivan S, Wordsworth S, et al. VIdeo assisted thoracoscopic lobectomy versus conventional Open LobEcTomy for lung cancer, a multi-centre randomised controlled trial with an internal pilot. The VIOLET Study. Available online: http://www.nets.nihr.ac.uk/projects/hta/130403
- Rocco G. Two lives. Eur J Cardiothorac Surg 2016;49:1541-2. [Crossref] [PubMed]
- Oparka J, Yan TD, Ryan E, et al. Does video-assisted thoracic surgery provide a safe alternative to conventional techniques in patients with limited pulmonary function who are otherwise suitable for lung resection? Interact Cardiovasc Thorac Surg 2013;17:159-62. [Crossref] [PubMed]
- Saji H, Inoue T, Kato Y, et al. Virtual segmentectomy based on high-quality three-dimensional lung modelling from computed tomography images. Interact Cardiovasc Thorac Surg 2013;17:227-32. [Crossref] [PubMed]
- Zaman M, Bilal H, Woo CY, et al. In patients undergoing video-assisted thoracoscopic surgery excision, what is the best way to locate a subcentimetre solitary pulmonary nodule in order to achieve successful excision? Interact Cardiovasc Thorac Surg 2012;15:266-72. [Crossref] [PubMed]
- Watanabe K, Nomori H, Ohtsuka T, et al. Usefulness and complications of computed tomography-guided lipiodol marking for fluoroscopy-assisted thoracoscopic resection of small pulmonary nodules: experience with 174 nodules. J Thorac Cardiovasc Surg 2006;132:320-4. [Crossref] [PubMed]
- Rocco G, Cicalese M, La Manna C, et al. Ultrasonographic identification of peripheral pulmonary nodules through uniportal video-assisted thoracic surgery. Ann Thorac Surg 2011;92:1099-101. [Crossref] [PubMed]
- Berry MF, D'Amico TA, Onaitis MW, et al. Thoracoscopic approach to lobectomy for lung cancer does not compromise oncologic efficacy. Ann Thorac Surg 2014;98:197-202. [Crossref] [PubMed]
- Liu CC, Shih CS, Pennarun N, et al. Transition from a multiport technique to a single-port technique for lung cancer surgery: is lymph node dissection inferior using the single-port technique?†. Eur J Cardiothorac Surg 2016;49 Suppl 1:i64-72. [PubMed]
- Pischik VG. Technical difficulties and extending the indications for VATS lobectomy. J Thorac Dis 2014;6:S623-30. [PubMed]
- Bertolaccini L, Rocco G, Viti A, et al. Geometrical characteristics of uniportal VATS. J Thorac Dis 2013;5 Suppl 3:S214-6. [PubMed]
- Ng CS, Rocco G, Wong RH, et al. Uniportal and single-incision video-assisted thoracic surgery: the state of the art. Interact Cardiovasc Thorac Surg 2014;19:661-6. [Crossref] [PubMed]
- Passera E, Rizzi A, Robustellini M, et al. Pulmonary aspergilloma: clinical aspects and surgical treatment outcome. Thorac Surg Clin 2012;22:345-61. [Crossref] [PubMed]
- Rocco G. VATS lung biopsy: the uni-portal technique. Multimed Man CardiothoracSurg 2005;2005:mmcts.2004.000356.
- Rocco G. Single port video-assisted thoracic surgery (uniportal) in the routine general thoracic surgical practice. Op Tech Stcvs 2009;14:326-35.
- Brunelli A, Xiume F, Refai M, et al. Bilateral staged uniportal VATS for synchronous lung cancers. Interact Cardiovasc Thorac Surg 2006;5:658-9. [Crossref] [PubMed]
- Paul S, Altorki NK, Sheng S, et al. Thoracoscopic lobectomy is associated with lower morbidity than open lobectomy: a propensity-matched analysis from the STS database. J Thorac Cardiovasc Surg 2010;139:366-78. [Crossref] [PubMed]
- Gonzalez-Rivas D, Paradela M, Fieira E, et al. Single-incision video-assisted thoracoscopic lobectomy: initial results. J Thorac Cardiovasc Surg 2012;143:745-7. [Crossref] [PubMed]
- Aragón J, Pérez Méndez I. From open surgery to uniportal VATS: asturias experience. J Thorac Dis 2014;6:S644-9. [PubMed]
- Wang BY, Tu CC, Liu CY, et al. Single-incision thoracoscopic lobectomy and segmentectomy with radical lymph node dissection. Ann Thorac Surg 2013;96:977-82. [Crossref] [PubMed]
- D'Amico TA. Long-term outcomes of thoracoscopic lobectomy. Thorac Surg Clin 2008;18:259-62. [Crossref] [PubMed]
- Rocco G, Martucci N, La Manna C, et al. Ten-year experience on 644 patients undergoing single-port (uniportal) video-assisted thoracoscopic surgery. Ann Thorac Surg 2013;96:434-8. [Crossref] [PubMed]
- Jutley RS, Khalil MW, Rocco G. Uniportal vs standard three-port VATS technique for spontaneous pneumothorax: comparison of post-operative pain and residual paraesthesia. Eur J Cardiothorac Surg 2005;28:43-6. [Crossref] [PubMed]
- Sihoe AD, Yim AP. Video-assisted pulmonary resections. In: Patterson GA, Cooper JD, Deslauriers J, et al. editors. Thoracic surgery (3rd edition). Philadelphia, USA: Elsevier, 2008:970-88.
- Harris CG, James RS, Tian DH, et al. Systematic review and meta-analysis of uniportal versus multiportal video-assisted thoracoscopic lobectomy for lung cancer. Ann Cardiothorac Surg 2016;5:76-84. [Crossref] [PubMed]
- Li WW, Lee TW, Lam SS, et al. Quality of life following lung cancer resection: video-assisted thoracic surgery vs thoracotomy. Chest 2002;122:584-9. [Crossref] [PubMed]
- Nagahiro I, Andou A, Aoe M, et al. Pulmonary function, postoperative pain, and serum cytokine level after lobectomy: a comparison of VATS and conventional procedure. Ann Thorac Surg 2001;72:362-5. [Crossref] [PubMed]
- Tamura M, Shimizu Y, Hashizume Y. Pain following thoracoscopic surgery: retrospective analysis between single-incision and three-port video-assisted thoracoscopic surgery. J Cardiothorac Surg 2013;8:153. [Crossref] [PubMed]
- Perna V, Carvajal AF, Torrecilla JA, et al. Uniportal video-assisted thoracoscopic lobectomy versus other video-assisted thoracoscopic lobectomy techniques: a randomized study. Eur J Cardiothorac Surg 2016;50:411-5. [Crossref] [PubMed]
Cite this article as: Passera E, Rocco G. From full thoracotomy to uniportal video-assisted thoracic surgery: lessons learned. J Vis Surg 2017;3:36.