Management for a complicated biliary stricture after iatrogenic bile duct injury
Introduction
Due to being a severe complication, bile duct injury (BDI) is still a challenging issue for biliary surgeons. Over the past decades, the literatures reported incidence of major BDI after laparoscopic cholecystectomy (LC) is about 0.3–0.5% (1-3). Recently, a prospective cohort study showed that complexity of referred BDI and rate of associated vascular injuries have increased over time (4). BDI may cause not only biliary stricture for cases without effective and timely management, even result in severe complications such as recurrent cholangitis and impairment to liver function, but also high morbidity and mortality, impaired quality of life, and substantial financial burdens to patients and society (1,5). Depending on the type of injury, optimal management of BDI is achieved with a multidisciplinary approach, including endoscopic, percutaneous, and surgical interventions (6). Here, we present a case of management for complicated biliary stricture after iatrogenic BDI.
Patient selection and workup
In March, 2014, a 69-year-old man was referred to us with a one-more-year history of recurrent jaundice and fever after LC. He underwent LC for cholecystolithiasis with chronic cholecystitis in November 2012. One month later, the man was re-admitted for severe jaundice. Considering the iatrogenic BDI caused by the primary operation (more information was unknown), he underwent Roux-en-Y choledochojejunostomy. Since then, he received several interventional measures, including percutaneous transhepatic biliary drainage (PTBD), percutaneous balloon balloon angioplasty, as well as percutaneous transhepatic cholangioplasty, with two biliary drainage tubes for left and right hepatic duct separately, trying to relieve repeated jaundice and high fever. However, the patient's condition had no significant improvement and even his weight loss of about 15 kilogram.
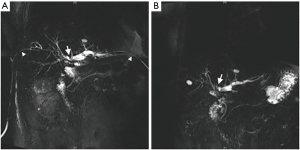
Pre-operative work-up included magnetic resonance cholangiopancreatography (MRCP) and computed tomographic angiography of the liver and biliary tract. MRCP showed stricture at hilar bile duct and dilatation of intrahepatic bile duct (Figure 1). Following a thorough discussion of treatment options and associated benefits and risks with the patient, we decided to proceed with a reconstruction of choledochojejunostomy. Risks and complications of the procedure included biliary leak, cholangitis, bleeding, anastomotic strictures, biliary cirrhosis, and the need for further surgeries.
Pre-operative preparation
Prior to the procedure, complete blood count, liver function test, coagulation function test, and cardiopulmonary function assessment were ordered (all normal) in addition to a full physical examination and patient history. Antibiotics were administered in that sequence no more than 30 minutes prior to the incision. Appropriate consent was obtained from the patient per our institutional protocol.
Equipment preference card
- Water knife;
- Bile duct probes;
- Fine catheter;
- 4–0 Vicryl suture;
- 5–0 Prolene suture;
- Self-retaining wound retractor.
Procedure
The operative procedure is demonstrated in Figure 2. The patient was placed in the supine position. After general anesthesia was induced, a Foley catheter was placed. Through the original incision, a down L-shaped incision below the right rib edge was made. Dense adherence was found in the area of biliary-enteric anastomosis, which was 40 cm away from enteric-enteric anastomosis. Part of the right and quadrate lobe of liver (about 4 cm × 4 cm) was marked and removed. Then, bile duct above the anastomosis was separated by water knife. After finding the original anastomosis, the bile duct was confirmed by a puncture. Part of the anterior wall of the bowel was incised. Anastomotic scar hyperplasia was significantly associated with stenosis. It was found that left and right hepatic was interrupted, and anastomosis of left hepatic duct almost closed.

Bile duct wall was separated from the liver tissue until opening the diameter of right hepatic duct to 2 cm, and the left to 2.5 cm. Due to the confluence of left and right hepatic duct to be adhered scar tissue, it was reshaped into an “8” shape with satisfaction. After exploration of the bile duct again, it was confirmed that the secondary bile duct was clear, and the hepatic duct of right posterior lobe was opening at the beginning of left hepatic duct. Two small stones, diameter of about 0.3 cm, was removed from bile duct, after cleaning with saline via fine catheter. After trimmed the scars of the anterior wall of the bowel, the bile duct jejunum anastomosis was sutured by 4–0 Vicryl and 5–0 Prolene intermittently. Hereafter, PTBD drainage tubes was removed. The outlet of left hepatic drainage tube was sutured with satisfaction. For the purpose of anti-reflux, the original intestinal anastomosis was suturing about 5 cm paralleled.
Total operative time was about 7 hours. Blood lose during the operation was about 200 mL. Two units of red blood cells and 200 mL of plasma was transfused Intraoperative.
Post-operative management
Following the procedure, the patient was admitted to the surgical intensive care unit for 24 hours observation and return to the regular wards. Postoperative evolution was uncomplicated and the patient was discharged on the 21st postoperative day. Two years after operation the patient was well and subsequent follow-up examinations did not reveal any symptoms of bile duct stricture.
Tips, tricks and pitfalls
- Treatment principles for BDI including restoring normal anatomical relationship of biliary system, retaining normal function of Oddi’s sphincter, maintaining the normal pathways of bile circulation, prevention of biliary stricture and reflux cholangitis. A reasonable individual treatment program should be developed according to the timing, location, scope and extent of BDI;
- If the BDI did not recognized until weeks after the primary operation, bile duct stricture might be formed due to repeated inflammation or multiple manipulations. Residual bile duct would become shorter, or even buried in the scar tissue;
- Due to the anatomical variation and deep location, right posterior hepatic duct more likely to be omitted. Preoperative PTBD angiography can not only contribute to the diagnosis, but also provide a guidance for intraoperative intubation via the PTBD sinus to facilitate finding the right hepatic duct. Ultrasound-guided puncture and intraoperative contrast-enhanced ultrasound, by which injection of contrast agent into the target bile duct to determine the drainage area of the bile duct, also would help to find the right hepatic duct;
- During dissecting hilar plate, which should be along the hilar to the right, the most difficult step is to expose and reshape the proximal bile duct. If necessary, removal of hepatic quadrate lobe would facilitate the search of intrahepatic bile duct. According to the actual situation, other ways also can be taken, such as incision of anterior transhepatic approach, as well as transverse portion of left hepatic duct approach;
- The key step of biliary tract reconstruction is dissection and reshape of hilar bile duct. Dissection should be along with the liver surface, via both left and right approach, to open the omentum hole. The method of “duct-to-mucosa” should be applied for biliary-enteric anastomosis as far as possible;
- The biliary supporting tube is not routinely placed, although it helps avoid the occurrence of anastomotic leakage and biliary stricture. We generally removed the tube 3 to 6 months post-operation, depending on intraoperative condition of the bile duct, the cholangiography and electronic choledochoscopy results after surgery, as well as general condition of the patient.
Discussion
A commonly accepted classification describing the type of BDI has not been available yet (8). Bismuth classification and Strasberg classification (an expansion of the Bismuth system), is currently the most widely used classification system for BDI. However, both of them still mainly based on the anatomical site of the BDI, with many limitations, such as not considering additional vascular involvement, and the type of injury factors (9). Moreover, no existing classification system takes into account all therapeutic and prognostic implications (10). Therefore, it is necessary to develop a new classification system according to the following conditions, such as not only anatomical site of the bile duct tree injury, but also type of injury factors, lesion characteristics, and prevention strategies.
Depending on the type of injury, percutaneous, endoscopic, and surgical interventions may account for the management of BDI. Percutaneous intervention is usually performed under ultrasound or radiological guidance, including abscess drainage, transhepatic biliary drainage, dilation of bile duct strictures and stent placement to maintain ductal patency, as well as management of complications from previous interventions. Although a randomized clinical trial showed that metallic stents should be considered an appropriate option in patients with benign biliary strictures and a bile duct diameter 6 mm or more (11). However, our experience does not recommend the use of metallic stents for biliary strictures in BDI patients because it does not improve the onset of recurrent cholangitis.
Surgical interventions are eligible for BDI patients with large lateral defects in major ducts, strictures refractory to percutaneous or endoscopic treatment, and nearly all complete transactions and ligations (6). Roux-en-Y hepaticojejunostomy is the best treatment option for most major BDI, and provides excellent long-term outcomes overall when the procedure is performed in tertiary referral center (2), or by an experienced hepatobiliary surgeon (10,12). It is worth noting that liver transplantation would be the last choice for BDI patients who developed biliary cirrhosis, and even liver failure (13).
The importance of timing of surgical repair for the outcome is controversial. It is still not clear whether the injury should be repaired immediately or a delayed repair is preferred. A retrospective study, 640 cases involved, was conducted to compare the results of immediate, early and late surgical repair for BDI sustained during a cholecystectomy. It was concluded that the best timing to repair a BDI was beyond 45 days (late repair group) and the best results could be expected in the hands of experienced hepatobiliary surgeons with bilio-enteric repair in the form of the Roux-en-Y hepaticojejunostomy (14). In another analysis of factors associated with successful surgical reconstruction, it was found that eradication of abdominal infection, complete cholangiography, use of correct surgical technique, and repair by an experienced biliary surgeon were the most important variables, whereas the timing of reconstruction was not independently significant (15).
For the case presented here, the patient underwent a second operation to reconstruct the biliary system in less than a month after the first LC, and resulted in severe post-operative complications. The patient did not achieve satisfactory outcomes till underwent the last surgical management more than one year after the original iatrogenic injury. This indicates that the choice of making an early repair is a difficult task and should be undertaken by an experienced hepatobiliary surgeon to offer the patient the best chance of a cure (16). Another retrospective analysis conducted by our team found that the timing of reconstruction depended on the downtrend of serum bilirubin levels and the control of inflammation, generally from 4 to 6 weeks after percutaneous or endoscopic interventions (17), which was similar to some other reports (18,19).
Some patients may develop complications of anastomotic and biliary strictures after Roux-en-Y hepaticojejunostomy for BDI. Reconstruction is often undergone after fully exposure of the anastomic area and assessment of the biliary anatomy with an electronic choledochoscopy if available. According to our experience, if strictures and inflammation are limited in the anastomic and extrahepatic area, a vertical incision and a horizontal suture are preferred in order to construct a 2–3 cm wide anastomosis. Moreover, a complete resection of strictures and a new hepaticojejunostomy are recommended, when those spreads to higher bile duct. Sometimes, dissection of the hilar plate or partial hepatectomy is needed to expose the normal bile duct (17). For this patient, considering extensive adhesion, complete resection of former anastomosis and a new Roux-en-Y hepaticojejunostomy was performed.
In some patients with complex BDI involving disruption of proximal bile duct (involving hepatic confluence) and injuries associated with concomitant vascular damage, partial hepatectomy is required (20). It has reported that two main groups of BDI patients require liver resection: those with an injury-induced liver necrosis necessitating early intervention, and those in whom hepatectomy is indicated for treatment of liver atrophy following long-term cholangitis (21). In our experiences, hepatectomy is not a standard procedure in surgical treatment of BDI. However, in some complex injuries, it should be considered to help the exposure of normal bile duct, even if there has no evidence on liver necrosis or lobar atrophy and fibrosis. According to literatures, hepatic resections in patients with BDI can be performed successfully with low mortality, although with significant morbidity, and with excellent long-term success (21). As showed in this case, removal of hepatic quadrate lobe would facilitate the search of intrahepatic bile duct.
Conclusions
It should be noted that biliary reconstruction for BDI is a challenging surgical procedure even in the hands of experienced hepatobiliary surgeons. Endoscopic and percutaneous interventions may be performed for definitive treatment or as adjuncts to surgical management. Only through comprehensive pre-operative evaluation and meticulous intraoperative manipulate, the technical challenge of reconstruction for complicated biliary stricture after BDI should be easily addressed.
Acknowledgements
Funding: This work was supported by grant from the National Natural Science Fund of China 81672925 (L Chen).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Flum DR, Cheadle A, Prela C, et al. Bile duct injury during cholecystectomy and survival in medicare beneficiaries. JAMA 2003;290:2168-73. [Crossref] [PubMed]
- Martin D, Uldry E, Demartines N, et al. Bile duct injuries after laparoscopic cholecystectomy: 11-year experience in a tertiary center. Biosci Trends 2016;10:197-201. [Crossref] [PubMed]
- Rystedt J, Lindell G, Montgomery A. Bile Duct Injuries Associated With 55,134 Cholecystectomies: Treatment and Outcome from a National Perspective. World J Surg 2016;40:73-80. [Crossref] [PubMed]
- Hogan NM, Dorcaratto D, Hogan AM, et al. Iatrogenic common bile duct injuries: Increasing complexity in the laparoscopic era: A prospective cohort study. Int J Surg 2016;33 Pt A:151-6.
- de Reuver PR, Sprangers MA, Rauws EA, et al. Impact of bile duct injury after laparoscopic cholecystectomy on quality of life: a longitudinal study after multidisciplinary treatment. Endoscopy 2008;40:637-43. [Crossref] [PubMed]
- Thompson CM, Saad NE, Quazi RR, et al. Management of iatrogenic bile duct injuries: role of the interventional radiologist. Radiographics 2013;33:117-34. [Crossref] [PubMed]
- Suo T, Chen L, Liu H, et al. Video demonstrating the reconstruction of choledochojejunostomy for biliary stricture after bile duct injury (BDI). Asvide 2017;4:107. Available online: http://www.asvide.com/articles/1415
- Lau WY, Lai EC. Classification of iatrogenic bile duct injury. Hepatobiliary Pancreat Dis Int 2007;6:459-63. [PubMed]
- Velidedeoglu M, Arikan AE, Uludag SS, et al. Clinical Application of Six Current Classification Systems for Iatrogenic Bile Duct Injuries after Cholecystectomy. Hepatogastroenterology 2015;62:577-84. [PubMed]
- Lau WY, Lai EC, Lau SH. Management of bile duct injury after laparoscopic cholecystectomy: a review. ANZ J Surg 2010;80:75-81. [Crossref] [PubMed]
- Coté GA, Slivka A, Tarnasky P, et al. Effect of Covered Metallic Stents Compared With Plastic Stents on Benign Biliary Stricture Resolution: A Randomized Clinical Trial. JAMA 2016;315:1250-7. [Crossref] [PubMed]
- Walsh RM, Henderson JM, Vogt DP, et al. Long-term outcome of biliary reconstruction for bile duct injuries from laparoscopic cholecystectomies. Surgery 2007;142:450-6; discussion 456-7. [Crossref] [PubMed]
- Leale I, Moraglia E, Bottino G, et al. Role of Liver Transplantation in Bilio-Vascular Liver Injury After Cholecystectomy. Transplant Proc 2016;48:370-6. [Crossref] [PubMed]
- Iannelli A, Paineau J, Hamy A, et al. Primary versus delayed repair for bile duct injuries sustained during cholecystectomy: results of a survey of the Association Francaise de Chirurgie. HPB (Oxford) 2013;15:611-6. [Crossref] [PubMed]
- Stewart L, Way LW. Laparoscopic bile duct injuries: timing of surgical repair does not influence success rate. A multivariate analysis of factors influencing surgical outcomes. HPB (Oxford) 2009;11:516-22. [Crossref] [PubMed]
- Kapoor VK. Bile duct injury repair: when? what? who? J Hepatobiliary Pancreat Surg 2007;14:476-9. [Crossref] [PubMed]
- Liu H, Shen S, Wang Y, et al. Biliary reconstruction and Roux-en-Y hepaticojejunostomy for the management of complicated biliary strictures after bile duct injury. Int Surg J 2015;2:179-86. [Crossref]
- Lillemoe KD, Melton GB, Cameron JL, et al. Postoperative bile duct strictures: management and outcome in the 1990s. Ann Surg 2000;232:430-41. [Crossref] [PubMed]
- de Reuver PR, Grossmann I, Busch OR, et al. Referral pattern and timing of repair are risk factors for complications after reconstructive surgery for bile duct injury. Ann Surg 2007;245:763-70. [Crossref] [PubMed]
- Jabłońska B. Hepatectomy for bile duct injuries: when is it necessary? World J Gastroenterol 2013;19:6348-52. [Crossref] [PubMed]
- Li J, Frilling A, Nadalin S, et al. Timing and risk factors of hepatectomy in the management of complications following laparoscopic cholecystectomy. J Gastrointest Surg 2012;16:815-20. [Crossref] [PubMed]
Cite this article as: Suo T, Chen L, Liu H, Ni X, Shen S, Wang Y, Liu H. Management for a complicated biliary stricture after iatrogenic bile duct injury. J Vis Surg 2017;3:33.


