Challenges in complex video-assisted thoracoscopic surgery and spontaneous respiration video-assisted thoracoscopic surgery procedures
Introduction
Since the development of the video-assisted thoracoscopic surgery (VATS) technique in 1992 (1,2), the field of thoracic surgery has developed at a rapid rate. These developments can be attributed to the exponential changes we have seen in technology, that have benefited surgeons by equipping centers with increasingly accurate technology for both the detection and removal of tumors. Without the invention of endoscopic instruments, many minimally invasive thoracic surgery procedures would not be possible.
Since the completion of the first video assisted procedure (1,2), surgeons have continuously developed the surgical skills and techniques to perform increasingly challenging procedures while at the same time expanded the indications for VATS procedures. The push for minimally invasive techniques had led to the development of many surgical tools and the innovation and completion of ever more complex operations. More recently, this minimally invasive trend has extended to include intubation and anesthesia as well.
Spontaneous respiration anesthesia (SRA) is a recent promising development in the field of minimally invasive surgery. It was not until 2004 that Pompeo et al. (3) began to explore this technique for thoracic procedures. Since 2011, we have performed more than 1,800 SRA procedures in our center ranging from wedge to sleeve resections.
In our hospital we complete nearly 40,000 surgical procedures each year with thoracic surgery accounting for about 3,000 of them. To achieve faster postoperative recovery of patients, we have been dedicated to the development of surgical skills that have allowed us to successfully complete many procedures under VATS that are complex even with open approach. Specifically, sleeve, trachea, and carina resections and reconstructions using either general or SRA techniques.
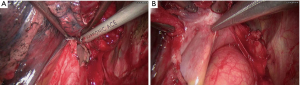
Challenges in double sleeve resections (Figure 1)

In 2007, our team performed our first complete VATS bronchial sleeve resection and reconstruction (5). To date we have completed more than 187 complete VATS sleeve resections including 149 bronchial sleeve resections, 34 double sleeve resections, and four superior vena cava (SVC) resection. Double sleeve resection is one of the most complicated of these sleeve resection procedures, specifically in regards to the anastomotic techniques and the demerits of a two-dimensional (2D) display system.
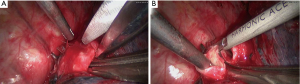
Contrary to traditional open approach, anastomosis under thoracoscopy can be challenging because of the limited angles for operation and frequent occurrence of thread winding. To overcome these angle limitations, a tailored ports design is predominant for a successful surgery (6). In our practice, an additional 5 mm incision can greatly ease surgical maneuvers and, in turn, shorten operative time. Wound protectors are also recommended, for they enable outstanding convenience in operation. Furthermore, our team uses a unique method to avoid the occurrence of thread winding in addition to running suture: Instead of pulling the other end of Prolene out of thoracic incision, we wind this end around small gauze with the needle tucked inside and leave the gauze far from surgical field (Figure 2).
Our center currently uses a glasses-free 3D thoracoscopic system in the majority of thoracic procedures. To take full advantage of its merits of improved depth and detail perception compared to the traditional 2D system, this glasses-free 3D thoracoscopic system has become a standard tool for any complicated procedures. Our use of a greatly facilitates every surgical maneuver especially during dissection and anastomosis of difficult operations. Moreover, we have also observed a shortened operative time of procedures completed using this system when compared with the traditional 2D display (7).
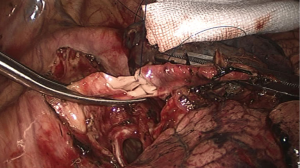
Challenges of endoscopic bronchoplasty, arterioplasty, and angioplasty of the SVC (Figure 3)

Our progressive experience and development in VATS reconstruction, such as bronchial sleeve reconstruction (5), half carinal reconstruction (9), double sleeve reconstruction (6), and tracheal and carinal reconstructions under general anesthesia (10), has recently led to a case experience of an endoscopic bronchoplasty, arterioplasty, and angioplasty of SVC for the radical treatment of right middle upper lung cancer (11). This patient was originally planned for a right upper lobe sleeve resection but intraoperative exploration revealed the mass to be located at the center of right upper lobe, protruding into the right main bronchus, the interlobar portion of right pulmonary artery (PA) and part of the lateral wall of SVC. Upon discussion with our team leader and other specialists at our institution, the decision to perform this ‘triple plasty’ procedure was made and successfully executed. Accurate intra-operative assessment is as vital as pre-operative imaging evaluation in the planning of such resections (12). There must be adequate length of PA and SVC proximally and distally to achieve vascular control. To make such assessment, it is important to open the pericardium to expose the SVC fully (Figure 4). In our case, it was also necessary to remove the tumour from the SVC first to get a better assessment of the extent of hilar involvement and dissect out the proximal PA. The other important learning point from this case was that the distal airway (Figure 5) may need to be dissected away from the distal PA to fully evaluate and obtain better vascular control of the PA when there is extensive tumour involvement.
Since the diameter of the SVC can be nearly half of the original width, to avoid high pressure on SVC after reconstruction, intravenous fluid replacement therapy should be slow. It may be helpful to place a subclavian vein catheter to the proximal part of SVC during induction of anaesthesia, to withdraw to the proximal part before blocking the SVC, and to place back into the distal part of SVC after reconstruction. Furthermore, ice cap for crania and intravenous administration of mannitol are recommended in countering the effects of cerebral venous return obstruction while blocking the SVC.
Challenges of SRA trachea and carina resections (Figure 6)

Though there is still debate among the thoracic community regarding SRA procedures, our experience has been consistently positive. Surprisingly, in our experience, SRA offers surgeons themselves advantages during one of the most complicated resection types: trachea and carina reconstructions.
Tracheal and carinal reconstructions are technically demanding in even traditional open operations. However, during such VATS procedures, the difficulty is further increased due to the loss of specific advantageous angles available during traditional open operations.
To date, we have successfully performed 16 cases of VATS tracheal and carinal reconstructions under SRA (awaiting publication). These patients adhered to the following criteria: (I) no significant defect in cardiac function, ECG, cardio ultrasound and coronary contrast computed tomography, is reported; (II) no remarkable airway secretion is observed; (III) body mass index (BMI) should be less than 25; (IV) sectional area of the tumor within trachea should be less than 75%. For those who are beyond this limit, a partial resection of the tumor with bronchoscopy is performed before the procedure; (V) no significant contraindications for epidural puncture are reported.
The anaesthesitic program for these procedures is epidural anesthesia and intravenous sedation with laryngeal-mask for spontaneous respiration. The patient is administered 0.3 mg of intravenous scopolamine. ECG, non-invasive blood pressure (NIBP), respiratory rate (RR) and Pulse Oxygen Saturation (SPO2) is monitored. While establishing fluid channel in right internal jugular vein and monitoring pathway in left radial artery, 4–5 L/min oxygen is given via nasal catheter. Patients are positioned in a left lateral decubitus position. T5–6 epidural puncture is performed, and then 3 mL of 2% lidocaine is injected into the epidural cavity. Patients are then repositioned to the dorsal decubitus position. Ten minutes’ observation to rule out potential total spinal anesthesia. Intravenous sedation is then conducted with 10 μg of Sufentanil and 2 μg of Propofol (TCI). When patients lose consciousness, a laryngeal mask is inserted for spontaneous respiration. 15 mL of 0.25% Ropivacaine was used for maintenance of anesthesia. Every 45–60 minutes, extra 5 mL of 0.25% Ropivacaine, 1–2 μg of Propofol (TCI) and 0.5–1 μg/kg of Dexemedetomidine is added for maintenance of anesthesia with bispectral index (BIS) around 40–60. After the incisions are made, the surgeon uses a 6–8 mL of a mixture of 2% lidocaine and 0.75% Ropivacaine is used for a vagus nerve block and 2% lidocaine is sprayed into the airway via laryngeal mask by the anesthetist with fiber bronchoscopy.
In the event of an emergency during operation, several catheters and instruments should be prepared: extended single lumen endobronchial tube (5.0# and 5.5#), cross-field enhanced endobronchial tube (5.0# and 5.5#), 5mm hollow rigid catheter for high frequent jet ventilation (HFJV), fiber bronchoscopy, HFJV ventilator and an extracorporeal membrane oxygenation (ECMO) machine. There are three major critical issues during SRA for tracheal and carinal reconstruction: management of airway, hypoxemia and hypercapnia. In management of airway, the predominate goal is to prevent asphyxia. Vagus nerve blocking and endotracheal spray of lidocaine can suppress significant choking which may lead to bleeding due to mishandling of vessel. Before opening the airway, absolute hemostasis of the operative field is required. After resection of the tracheal/bronchial tumor, the distal opening of the airway is suspended and any fluid within operative field is sucked out to prevent aspiration. Preserving the left pleural membrane intact while mobilizing the carina plays a key role in preventing hypoxemia. Before attempting intubation for hypoxemia however, several interventions are available: 10L/min continuous oxygen flow into airway via laryngeal mask and extra HFJV into right lung via 5mm hollow rigid catheter. In our practice, with normal SpO2, permissive hypercapnia is safe as long as the PaCO2 slowly rises to around 75–110 mmHg.
Conclusions
There is an increasing body of evidence indicating the importance of higher volume experience on surgical outcomes. Many countries in Asia have large and/or concentrated populations. As China is the most populated country in the world, it is no wonder that specialized centers around the country, particularly in the large cities, complete an astounding number of surgical procedures each year. Our hospital specializes in thoracic disease and, as a result, thoracic surgery accounts for the largest percentage of surgical procedures performed in our high volume center.
The most significant disadvantage for any of the above surgical procedures is the need for a highly experienced surgical team. Our long term high volume thoracic experience has equipped us with a talented multidisciplinary team with the ability to confidently and safely perform many types of complicated VATS procedures. However, for a number of surgical centers worldwide, the learning curve for such procedures leaves them at a significant disadvantage and perhaps even inability to perform these surgeries.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Lewis RJ, Caccavale RJ, Sisler GE, et al. One hundred consecutive patients undergoing video-assisted thoracic operations. Ann Thorac Surg 1992;54:421-6. [Crossref] [PubMed]
- 2Davies AL, Panasuk DB. Video-assisted thoracic surgery: our first 20 cases. Del Med J 1992;64:267-72. [PubMed]
- Pompeo E, Mineo D, Rogliani P, et al. Feasibility and results of awake thoracoscopic resection of solitary pulmonary nodules. Ann Thorac Surg 2004;78:1761-8. [Crossref] [PubMed]
- Li J, Liu H, He J, et al. Video-assisted thoracoscopic surgery (VATS) double sleeve resection of left upper lung cancer. Asvide 2017;4:101. Available online: http://www.asvide.com/articles/1409
- Shao WL, Liu LX, He JX, et al. Bronchial sleeve resection and reconstruction of pulmonary artery by video-assisted thoracic small incision surgery for central lung cancer: a report of 139 cases. Zhonghua Wai Ke Za Zhi 2007;45:1530-2. [PubMed]
- Huang J, Li J, Qiu Y, et al. Thoracoscopic double sleeve lobectomy in 13 patients: a series report from multi-centers. J Thorac Dis 2015;7:834-42. [PubMed]
- Yang C, Mo L, Ma Y, et al. A comparative analysis of lung cancer patients treated with lobectomy via three-dimensional video-assisted thoracoscopic surgery versus two-dimensional resection. J Thorac Dis 2015;7:1798-805. [PubMed]
- Li J, Liu H, He J, et al. Video-assisted thoracoscopic surgery (VATS) triple plasty for resection of right middle upper lung cancer. Asvide 2017;4:102. Available online: http://www.asvide.com/articles/1410
- Xu X, Chen H, Yin W, et al. Thoracoscopic half carina resection and bronchial sleeve resection for central lung cancer. Surg Innov 2014;21:481-6. [Crossref] [PubMed]
- He J, Wang W, Li J, et al. Video-assisted thoracoscopic surgery tracheal resection and carinal reconstruction for tracheal adenoid cystic carcinoma. J Thorac Dis 2016;8:198-203. [PubMed]
- Shao W, Yin W, Wang W, et al. Glasses-free three-dimensional endoscopic bronchoplasty, arterioplasty, and angioplasty of the superior vena cava for the radical treatment of right middle upper lung cancer. J Thorac Dis 2016;8:608-11. [Crossref] [PubMed]
- Xu X, Chen H, Yin W, et al. Initial experience of thoracoscopic lobectomy with partial removal of the superior vena cava for lung cancers. Eur J Cardiothorac Surg 2015;47:e8-e12. [Crossref] [PubMed]
- Li J, Liu H, He J, et al. Video-assisted thoracoscopic surgery (VATS) spontaneous respiration anesthesia (SRA) carinal resection and reconstruction. Asvide 2017;4:103. Available online: http://www.asvide.com/articles/1411
Cite this article as: Li J, Liu H, Liu J, Zou X, Mo L, Pan H, Peng G, Shao W, Hamblin L, Liang L, Dong Q, He J. Challenges in complex video-assisted thoracoscopic surgery and spontaneous respiration video-assisted thoracoscopic surgery procedures. J Vis Surg 2017;3:31.