The Shanghai Pulmonary Hospital uniportal subxiphoid approach for lung segmentectomies
Introduction
According to recent meta-analyses lung segmentectomy may be considered an oncologic equivalent treatment to lobectomy for non-small cell lung cancer (NSCLC) sized 20 mm or smaller (1,2). The benefit of segmentectomy over lobectomy is the relative sparing of healthy lung tissue and the avoidance of potentially significant reductions in lung function post-operatively.
This not only reduces post-operative morbidity in otherwise healthy patients but also makes surgical resection and potential cure possible in patients with poorer baseline lung function who would not be suitable for lobectomy.
Minimally invasive lung resections reduce surgical trauma and consequently have shorter recovery times and lower morbidity than traditional open-techniques. Intercostal uniportal video-assisted thoracoscopic surgery (VATS) approaches have been shown to offer additional benefits over multiportal VATS including reduced rates of acute and chronic pain, allodynia and hypaesthesia (3). Uniportal subxiphoid VATS (SVATS) avoids the intercostal neurovascular bundle and does not involve any rib-spreading and therefore theoretically reduces the risk of post-operative morbidity compared to intercostal VATS approaches. SVATS also allows the resection of bilateral lesions in a single procedure. The results of the Shanghai Pulmonary Hospital experience showed the feasibility and the safety of the subxiphoid approach for lobectomies (4-6).
The aim of this article is to present a summary of the Shanghai Pulmonary Hospital’s experience of SVATS segmentectomy, including feasibility, indications and complications in benign and malignant disease.
Indications for SVATS segmentectomy
The surgical indications included:
- Primary stage Ia–b lung cancer, with ground glass opacity (GGO) of ≤2.5 cm and consolidation ≤1.5 cm;
- Oligometastases ≤3 nodules;
- Selected cases of localised infectious lung disease or benign lung tumor discussed within our multidisciplinary team prior to surgery.
Exclusion criteria included:
- Patients who had lesions located within the inner third of the lung field;
- Diffuse dense adhesions;
- Lung cancer with lymph node enlargement;
- Body mass index, BMI >30;
- Cardiomegaly patients with left-side lesions;
- Target bronchus surrounded by calcified lymph nodes;
- Reoperation.
Operating room set up and surgical instruments
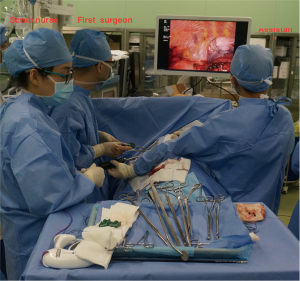
The standard set-up of the operating room consisted of a single monitor, positioned cranially, above the head of the patient. The first surgeon is positioned on the ventral side of the patient with the assistant surgeon and the scrub nurse on the dorsal side (Figure 1).
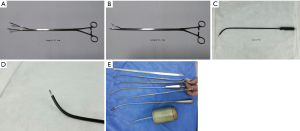
A 10-mm, 30° angled HD video-thoracoscope was used in all cases. Widely available VATS instruments plus specially-designed SVATS instruments [Shanghai Medical Instruments (Group) Ltd.] were also used (Figure 2).
Operative technique
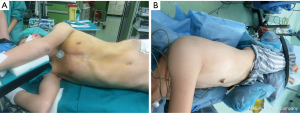
Positioning of the patients and anesthetic technique (Figure 3)
After induction of general anaesthesia and intubation with a double-lumen endotracheal tube, patients are placed in the lateral decubitus position (target side up) with a backward inclination of 30°
Left-sided resection via a SVATS approach often involves applying pressure to the pericardium which may result in arrhythmias and potential circulatory instability. For these patients, a central-venous catheter was placed at the start of the procedure for use in these circumstances.
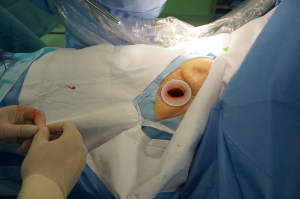
Creation of the port (Figure 4)
If the infrasternal angle is within normal limits (≥70o), a 4cm-long horizontal subxiphoid incision is made. A 4-cm longitudinal incision is used in patients with an infrasternal angle less than 70°. Blunt dissection of the subcutaneous tissue exposes the rectus abdominis muscle and its fibers are divided longitudinally to expose the xiphoid process. The xiphoid process is then completely excised to improve access and avoid the need for sternal retraction. A retrosternal tunnel is created by blunt, index-finger, dissection and a wound protector inserted. Under thoracoscopic visualization the pleura is opened and the pericardial fat tissue removed using a specially-designed long electrocautery blade. This instrument allows elevation, dissection and cauterization of the tissues and is used throughout the operation (Figure 2).
Basic surgical principles (Figure 5)
The camera is maintained in the caudal portion of the port leaving the cephalic section free for insertion and maneuvering the other surgical instruments.
Before any lung resection is performed, the whole ipsilateral chest cavity is inspected to exclude any significant adhesions or any progression of disease/additional pathology not seen on pre-operative imaging. Throughout this inspection, and the ongoing operation, the lung is retracted with specifically-designed lung graspers who optimize hilar exposure whilst reducing potential interference with the other instruments.
A long, metal, sucker with a distal curvature may be used carefully to displace the heart medially and so reduce any interference from the beating heart on the other VATS instruments. This is particularly useful in left-sided procedures but extra caution must be taken to reduce the previously mention risk of subsequent arrhythmias.
Performing the segmentectomies
The target vessels, fissures and bronchus are exposed and divided sequentially, using appropriate endostaplers. When amenable, surgical endoclips or a variety of energy-based devices where used to transect minor pulmonary arteries, divides thin portions of a fissure and during lymph node excision.
Lymph nodes
Initially, only lymph node sampling was performed during the SVATS segmentectomy procedures. However, with increasing experience, we were able to improve the technique and are now able to perform a systematic lymph node dissection with removal of at least 3 N stations (as per the IASLC/Mountain classification).
Conversion to open
If required, the operation may be converted to a multiportal VATS procedure with the addition of an extra port, usually in the 5th intercostal space, between the anterior and the mid-axillary lines. If there were multiple, significant adhesions or other major concerns intra-operatively, the procedure was converted to open using a muscle-sparing anterolateral thoracotomy at the level of the 5th intercostal space.
Closure and post-operative management
At the end of the procedure, one drain is placed through the subxiphoid incision and a thin percutaneous drain is placed under thoracoscopic-vision at the level of the 8th intercostal space.
After surgery, the patient is transferred to an intensive post-operative care unit for one night and then to the normal surgical ward. Early mobilization is important and the physiotherapists were extensively involved as part of an enhanced-recovery policy.
The chest drains were removed when there was no air-leakage and the fluid drainage was less than 300 mL in 24 hours. Patients were normally discharged the day after chest-drain removal and reviewed ten days later in the outpatient clinic.
Video atlas of uniportal SVATS segmentectomy
Right segmentectomies
Right upper lobe segmentectomies
- Apical S1 (Figure 6);
- Posterior S2 (Figure 7);
- Anterior S3 (Figure 8);
- Apicoanterior S1, S3 (Figure 9);
- Apicoposterior S1, S2 (Figure 10).





Right middle lobe segmentectomies


Right lower lobe segmentectomies
- Superior S6 (Figure 13);
- Basal S7–S10 (Figure 14);
- Medio-basal S7 (Figure 15);
- Antero-latera-basal S8, S9 (Figure 16).




Left segmentectomies
Left upper lobe segmentectomies
- Apicoanterior S1, S3 (Figure 17);
- Apicoposterior S1, S2 (Figure 18);
- Anterior S3 (Figure 19);
- Trisegmentectomy S1–S3 (Figure 20);
- Lingulectomy S4, S5 (Figure 21).





Left lower lobe segmentectomies
- Basal (Figure 22).

Results
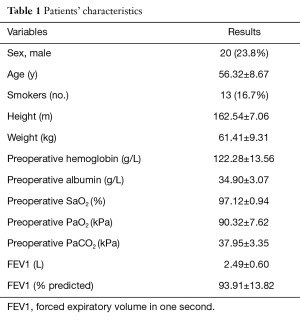
A total of 79 consecutive patients underwent 84 subxiphoid segmentectomies for malignant or benign pulmonary diseases between September 2014 and January 2016. Twenty patients were male and 59 were female. The mean age was 56.32 years (range, 36−79 years). Within the group, four patients underwent bilateral segmentectomies and one patient underwent unilateral double segmentectomies.
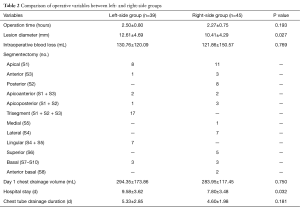
For analysis, the patients were divided into two groups depending on operative side. There were 45 segmentectomies in right-side group and 39 segmentectomies left-side group. The mean operation time was 2.38±0.77 hours, and mean operative blood loss was 126.09±136.17 mL. characteristics are detailed in Tables 1 and 2. Patients in left-side group had a larger mean lesion diameter (12.61±4.69 mm), compared to right-side patients (10.41±4.29 mm) (P<0.05). Patients in the left-side group also had significantly longer-in patient stays post-operatively. However, operative time, intra-operative blood loss and duration of chest drain use were equal across the two groups.
Full table
Full table
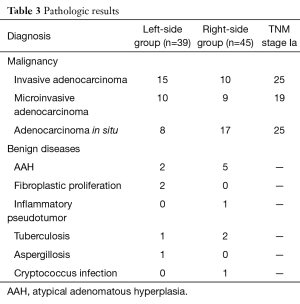
Pathological examination showed adenocarcinoma was the most common malignancy, including invasive, micro-invasive and adenocarcinoma in situ. The incidence of benign diseases was lower, including atypical adenomatous hyperplasia (AAH), inflammatory pseudotumour, tuberculosis, aspergillosis, and cryptococcus infection (Table 3).
Full table
Clinical and operative results
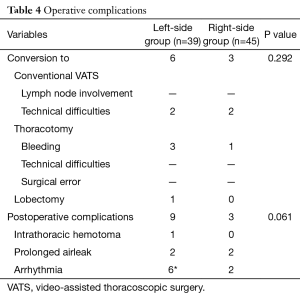
Nine segmentectomies (10.7%) had an intra-operative change of procedure as detailed in Table 4. Four were converted to thoracotomy, and of these one had a lobectomy rather than a segmentectomy. Four were converted to conventional VATS lobectoctomy. A total of 12 patients (15.1%) had postoperative complications, including 1 haematoma, 4 prolonged air-leaks and 8 arrhythmias. There was no significant difference in postoperative complication rates between the two groups.
Full table
Comment
We have presented our technique for SVATS segmentectomy which has been developed and adapted throughout this case series. Previous studies have shown that segmentectomy is oncologically equivalent to lobectomy for small malignant lesions and this paper suggests that the minimally invasive SVATS approach is a feasible option for unilateral and bilateral disease.
In the presented series that included our learning curve period we have experienced relative low conversion rates and few complications post-operatively and believe that, SVATS segmentectomy is a good option for many patients.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study has been approved by the institutional ethical committee and an informed consent was obtained from every patient. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
References
- Bao F, Ye P, Yang Y, et al. Segmentectomy or lobectomy for early stage lung cancer: a meta-analysis. Eur J Cardiothorac Surg 2014;46:1-7. [Crossref] [PubMed]
- Zhang L, Li M, Yin R, et al. Comparison of the oncologic outcomes of anatomic segmentectomy and lobectomy for early-stage non-small cell lung cancer. Ann Thorac Surg 2015;99:728-37. [Crossref] [PubMed]
- Hirai K, Takeuchi S, Usuda J. Single-incision thoracoscopic surgery and conventional video-assisted thoracoscopic surgery: a retrospective comparative study of perioperative clinical outcomes†. Eur J Cardiothorac Surg 2016;49 Suppl 1:i37-41. [PubMed]
- Aresu G, Wu L, Lin L, et al. The Shanghai Pulmonary Hospital subxiphoid approach for lobectomies. J Vis Surg 2016;2:135. [Crossref]
- Song N, Zhao DP, Jiang L, et al. Subxiphoid uniportal video-assisted thoracoscopic surgery (VATS) for lobectomy: a report of 105 cases. J Thorac Dis 2016;8:S251-7. [PubMed]
- Hernandez-Arenas LA, Lin L, Yang Y, et al. Initial experience in uniportal subxiphoid video-assisted thoracoscopic surgery for major lung resections. Eur J Cardiothorac Surg 2016. [Epub ahead of print]. [Crossref] [PubMed]
- Aresu G, Weaver H, Jiang L, et al. Right upper lobe apical segmentectomy S1. Asvide 2016;3:468. Available online: http://www.asvide.com/articles/1243
- Aresu G, Weaver H, Jiang L, et al. Right upper lobe posterior segmentectomy S2. Asvide 2016;3:469. Available online: http://www.asvide.com/articles/1244
- Aresu G, Weaver H, Jiang L, et al. Right upper lobe anterior segmentectomy S3. Asvide 2016;3:470. Available online: http://www.asvide.com/articles/1245
- Aresu G, Weaver H, Jiang L, et al. Right upper lobe apico-anterior segmentectomy S1, S3. Asvide 2016;3:471. Available online: http://www.asvide.com/articles/1246
- Aresu G, Weaver H, Jiang L, et al. Right upper lobe apico-posterior segmentectomy S1, S2. Asvide 2016;3:472. Available online: http://www.asvide.com/articles/1247
- Aresu G, Weaver H, Jiang L, et al. Right middle lobe medial segmentectomy S4. Asvide 2016;3:473. Available online: http://www.asvide.com/articles/1248
- Aresu G, Weaver H, Jiang L, et al. Right middle lobe lateral segmentectomy S5. Asvide 2016;3:474. Available online: http://www.asvide.com/articles/1249
- Aresu G, Weaver H, Jiang L, et al. Right lower lobe superior segmentectomy S6. Asvide 2016;3:475. Available online: http://www.asvide.com/articles/1250
- Aresu G, Weaver H, Jiang L, et al. Right lower lobe basal segmentectomy S7–S10. Asvide 2016;3:476. Available online: http://www.asvide.com/articles/1251
- Aresu G, Weaver H, Jiang L, et al. Right lower lobe medio-basal segmentectomy S7. Asvide 2016;3:477. Available online: http://www.asvide.com/articles/1252
- Aresu G, Weaver H, Jiang L, et al. Right lower lobe antero-lateral-basal segmentectomy S8, S9. Asvide 2016;3:478. Available online: http://www.asvide.com/articles/1253
- Aresu G, Weaver H, Jiang L, et al. Left upper apicoanterior segmentectomy S1, S3. Asvide 2016;3:479. Available online: http://www.asvide.com/articles/1254
- Aresu G, Weaver H, Jiang L, et al. Left upper apicoposterior segmentectomy S1, S2. Asvide 2016;3:480. Available online: http://www.asvide.com/articles/1255
- Aresu G, Weaver H, Jiang L, et al. Left upper anterior segmentectomy S3. Asvide 2016;3:481. Available online: http://www.asvide.com/articles/1256
- Aresu G, Weaver H, Jiang L, et al. Left upper trisegmentectomy S1–S3. Asvide 2016;3:482. Available online: http://www.asvide.com/articles/1257
- Aresu G, Weaver H, Jiang L, et al. Left upper lingulectomy S4, S5. Asvide 2016;3:483. Available online: http://www.asvide.com/articles/1258
- Aresu G, Weaver H, Jiang L, et al. Left lower basal segmentectomy. Asvide 2016;3:484. Available online: http://www.asvide.com/articles/1259
Cite this article as: Aresu G, Weaver H, Wu L, Lin L, Jiang G, Jiang L. The Shanghai Pulmonary Hospital uniportal subxiphoid approach for lung segmentectomies. J Vis Surg 2016;2:172.