Single port video-assisted thoracoscopic thymectomy
Introduction
Over the past decade, video-assisted thoracic surgery (VATS) has started to replace median sternotomy for the resection of non-invasive anterior mediastinal masses, including thymoma. The first thoracoscopic approaches for thymectomy were reported in 1993 by Sugarbaker (1) and by a Belgian group (2). Since then, the minimally invasive approach has become the preferred and standard operation for the treatment of thymic disease, whenever possible. In fact, many studies confirmed that, compared to standard sternotomy, VATS thymectomy results in less post-operative pain, better preserved pulmonary function, improved cosmesis (which can be particularly important to many young female myasthenia gravis patients) and is oncologically feasible for non-invasive thymomas as long as en bloc resection of the tumour is achieved (3-6).
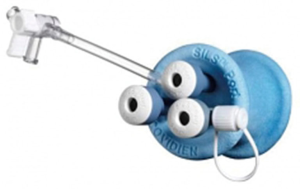
Most published reports regarding this procedure have focused on the right-sided approach, which has been adopted by most surgeons as the space in the right chest cavity is relatively large, with little interference from the heart, and the superior vena cava acts as an anatomical landmark. Classically three ports are used, but the current trend is to reduce the number of ports and minimize the length of incisions to further decrease postoperative pain, chest wall paraesthesia, and length of hospitalization. Uniportal VATS thymectomy for non-invasive mediastinal tumour resection is proven to be a safe and reliable method (7,8). In this paper we describe and illustrate our technique for single port VATS thymectomy. To facilitate the dissection, we adopted the use of a singular access device [single incision laparoscopic surgery (SILS) port, Covidien] that permits the insertion of three or four instruments through a right-sided single 3-cm incision together with CO2 insufflation, without rib spreading.
Patient selection
Uniportal VATS thymectomy for thymoma should be generally confined to small (
Surgical technique
Preparation
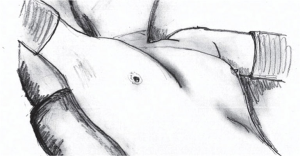
The patient is positioned in a 30-degree semi-supine position with a roll placed under the right shoulder and the ipsilateral arm is held abducted over a padded L-shaped bar to expose the axilla (Figure 1). The pulse oximeter is placed on the right hand fingers to check for signs of potential ischemia due to the bandages being too tight. The left arm is held extended on a padded board, so access to the left side is possible if required. The right side is the preferred approach, as there is more room for manipulation with the heart out of the way, and the anatomical landmarks (phrenic nerve, superior vena cava and innominate vein) are easily identifiable. The left-sided approach is used for thymomas located exclusively on that side. The surgeon and the assistant stand on the same side while the scrub nurse stands on the opposite side.
Procedure
A right-sided 3 cm lateral muscle-sparing incision is made at the fifth intercostal space. The single incision laparoscopic surgery (SILS) port device (Figure 2) is then inserted troughs the intercostal space without rib spreading (Figure 3). When the device is perfectly adherent to the skin, the insufflation of CO2 with a 6 L/min flow and 8 mmHg pressure is started. CO2 insufflation is used to help collapse the lung and to facilitate the dissection of the fat plane and it is continued throughout the procedure. We then introduce through the port a 30° 5-mm camera for inspection of the mediastinum and confirmation that the lesion is resectable. Usually there is no need for any lung retraction. The right phrenic nerve is seen coursing superior and lateral on the superior cava vein, the internal mammary artery vein and artery are also easily identifiable. In all cases the dissection starts from the pericardial fat plane and then is carried out superiorly to identify the anonymous vein. We always use the LigaSure device (5-mm blunt 37 lengths, Covidien) for tissue dissection and small vessel division. Dissection starts from the inferior border at the pericardial reflection, proceeding lateral to medial across the midline. When the left pleura and left phrenic nerve are reached, the dissection is directed cranially, parallel to the nerve. Once the pleural dissection has been completed, the thymus is then mobilized and retracted laterally and dissected off of the underlying pericardium. The bilateral upper gland is then stripped down from the neck in order to reveal the thymic vein (Figure 4). The removed thymus and fat tissue is finally placed in a specimen bag and taken out. At the end of the procedure, we introduce a 28 French chest tube through the same incision.


Tips, tricks and pitfalls
From our experience with uniportal VATS, we encountered several strategies that helped minimize the risk of capsular breakage and tumour seeding. Well-encapsulated tumours screened on thoracic computed tomography scan are ideal for the uniportal approach. We always perform a magnetic resonance image of the chest to identify the content of the lesion (cystic versus solid) and potential involvement of surrounding structures.
Tumours >4-mm in diameter and radiological suspicion of brachiocephalic vein invasion should be excluded from the uniportal approach.
The tumour should be approached from the ipsilateral side (right/left approach) so that dissection is performed under direct vision. However, for midline masses, we always recommend the right-sided approach.
The tumour should be dissected last, using a no-touch technique. The non-tumourous part of the gland is always dissected first and used for grasping and traction when dissecting the tumour, thus avoiding touching or grasping the tumour capsule. The use of the SILS port device is “effective” and safe and facilitates mediastinal mass resection without the need for extra incisions. Dedicated instruments such as the Kymerax Precision-Drive Articulating Surgical System (robotic instrument enabled precise instrument articulation and control) can be used to increase ergonomy and reach within the chest. This robotic aid should be considered for more complex procedures, but we do not consider it necessary for localized lesions.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Sugarbaker DJ. Thoracoscopy in the management of anterior mediastinal masses. Ann Thorac Surg 1993;56:653-6. [Crossref] [PubMed]
- Coosemans W, Lerut TE, Van Raemdonck DE. Thoracoscopic surgery: the Belgian experience. Ann Thorac Surg 1993;56:721-30. [Crossref] [PubMed]
- Ng CS, Wan IY, Yim AP. Video-assisted thoracic surgery thymectomy: the better approach. Ann Thorac Surg 2010;89:S2135-41. [Crossref] [PubMed]
- Rückert JC, Walter M, Müller JM. Pulmonary function after thoracoscopic thymectomy versus median sternotomy for myasthenia gravis. Ann Thorac Surg 2000;70:1656-61. [Crossref] [PubMed]
- Cheng YJ, Kao EL, Chou SH. Videothoracoscopic resection of stage II thymoma: prospective comparison of the results between thoracoscopy and open methods. Chest 2005;128:3010-2. [Crossref] [PubMed]
- Scarci M, Pardolesi A, Solli P. Uniportal video-assisted thoracic surgery thymectomy. Ann Cardiothorac Surg 2015;4:567-70. [PubMed]
- Wu CY, Heish MJ, Wu CF. Single port VATS mediastinal tumour resection: Taiwan experience. Ann Cardiothorac Surg 2016;5:107-11. [Crossref] [PubMed]
- Wu L, Lin L, Liu M, et al. Subxiphoid uniportal thoracoscopic extended thymectomy. J Thorac Dis 2015;7:1658-60. [PubMed]
- Bedetti B, Solli P, Lawrence D, et al. Introduction of the single incision laparoscopic surgery (SILS) port device. Asvide 2016;3:352. Available online: http://www.asvide.com/articles/1121
- Bedetti B, Solli P, Lawrence D, et al. Uniportal video-assisted thoracic surgery (VATS) thymectomy. Asvide 2016;3:353. Available online: http://www.asvide.com/articles/1122
Cite this article as: Bedetti B, Solli P, Lawrence D, Panagiotopoulos N, Hayward M, Scarci M. Single port video-assisted thoracoscopic thymectomy. J Vis Surg 2016;2:149.