Role of thoracoscopy, mediastinoscopy and laparoscopy in the diagnosis and staging of malignant pleural mesothelioma
Introduction
Malignant pleural mesothelioma (MPM) is a highly aggressive malignancy. The overall prognosis of MPM patients is poor and first line doublet chemotherapy (cisplatin + alimta) only marginally extends survival (1,2). Majority of MPM patients are diagnosed in advanced disease stages and therefore are not considered for surgery. Nevertheless, for a selected group of patients with epithelioid histology and an early disease stage multimodal therapy, comprised of cyto-reductive surgery in combination with intraoperative heated chemotherapy and with or without postoperative chemotherapy and radiation therapy, may be a viable option in peruse of life extension (3-7). Notably however, this aggressive therapeutic approach is associated with substantial morbidity and mortality rates and optimal patient selection is mandatory in order to maximize the potential benefits of surgery based multimodality therapeutic (MMT) (3-6). Given that clinical staging of MPM, especially in early disease stages, is often inaccurate in predicting the pathological stage of disease and taking into consideration that large tissue samples are frequently needed in order to establish a definite diagnosis of MPM histologic subtype we advocate that minimally invasive surgical sampling and staging of MPM patient is an important step in the workup of patients considered for surgery-based MMT (8-10). In this report we summarize the benefits of surgical sampling and staging of MPM patients prior to definite surgical resection. The complete clinical workup of this group of patients prior to surgery is beyond the scope of this concise review.
Benefits of thoracoscopy in confirming the diagnosis and subtype of MPM
MPM is generally classified into three histologic subtypes: epithelioid, sarcomatoid and mixed. Epithelioid MPM is the predominant type (50–60% of cases) and is associated with the best prognosis (10). Since the histologic appearance of MPM can greatly vary even within the same patient and since other malignant and benign conditions may mimic its appearance, it is often impossible to ascertain a concrete diagnosis of MPM and its subtype unless sufficient amounts of tissue are available for analysis (10). Consequently, it is not surprising that cytological analysis of pleural effusions as well as histological analysis of fine needle aspirates is frequently interpreted as suspicious for MPM but not as confirmatory of the diagnosis. While imaging guided needle biopsies have shown some promise in improving MPM diagnostics, thoracoscopy directed sampling of suspicious foci in the pleura remains the most sensitive method to obtain MPM diagnosis. This is especially true in early stages of disease when pleural thickening is only limited and recurrent pleural effusions are the most prominent manifestation of disease (10).
Benefits of thoracoscopy in assessing resectability of MPM
In cases where suspicion for mesothelioma is present and prior less invasive methodologies have failed to confirm a diagnosis, patients are taken to the operating room for an exploratory video assisted thoracoscopy. In addition to adequate tissue sampling several important characteristics of the disease can be evaluated during this procedure. Specifically the degree to which the tumor involves the chest wall, diaphragmatic and pericardial surfaces can be determined and, the extent to which the tumor infiltrates the visceral pleural and inter-lobar fissures can be defined. This valuable data helps the surgeon assess whether during definitive surgery the lung can be spared and whether resection of the pericardium and diaphragm may be required. Once these observations and the pre-surgical imaging indices of tumor invasion to chest wall and visceral organs are evaluated as a whole, a decision can be made with respect to the resectability of the tumor and with respect to the operative procedure of choice [pleurectomy and decortication (PD) or extrapleural pneumonectomy (EPP)].
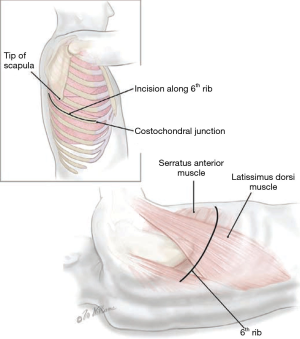
In our practice, for patients with a presumed diagnosis based on pleural fluid analysis or fine needle aspiration, we perform a diagnostic thoracoscopy to confirm the diagnosis. However, we do not perform a diagnostic thoracoscopy for patients that already have an established diagnosis of MPM based on a review of outside thoracoscopic or open pleural biopsy specimens unless the information regarding the extent of visceral pleural involvement will affect our overall decision to recommend surgery. This may be the case in patients that may tolerate PD but not EPP. If VATS is performed, we recommend placing the surgical incisions along the incision line that would eventually be placed during the definite surgery. This may facilitate future resection of these sites if seeded by tumor cells (Figure 1). Pleurodesis may be performed at time of VATS for patients that are not considered for surgery. However, if a cyto-reductive surgery is considered we do not perform pleurodesis since this may complicate the extrapleural dissection.
Benefits of mediastinoscopy in staging N2 nodes
In MPM the sensitivity of PET-CT for staging of the mediastinum is limited and in as much as 30% of cases pathological upstaging of the N category eventually occurs (8,11-13). This is of particular importance when considering N2 disease, as common practice is to recommend initial chemotherapy for patients with N2 disease while surgery may be offered to patients with N0 or N1 disease. Further, based on the proposed adjustments to the pathologic staging of epithelial MPM it is evident that diseases spread to station 2 and 4 nodes predict worse prognosis relative to disease spread to station 5 to 9 nodes (14). Thus, optimal staging of the mediastinum is desired in potential surgical candidates. Mediastinoscopy offers an attractive and safe approach in this regard.
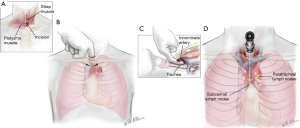
The surgical incision site, major anatomic structures to consider and lymph node to sample during mediastinoscopy are shown in Figure 2. Dissection of the tracheal plane, sampling of lymph nodes at station R4 and hemostasis are shown in the short video (Figure 3).

Benefits of laparoscopy in assessing transdiaphragmatic extension and peritoneal seeding
MPM trans-diaphragmatic disease invasion and/or disease spread to the peritoneum is missed in up to 10% of cases by conventional imaging modalities (CT scan, PET-CT or MRI) (8,11-13). Thus, prior to performing an aggressive cyto-reductive surgery we routinely explore the peritoneum. If the peritoneum is clear to eye inspection, we perform random biopsies and also wash the peritoneum for cytology.
Summary
Published series indicate that minimally invasive surgical staging of MPM patients prior to definite surgical resection has resulted in change of plan in up to 25% of cases thus highlighting the importance of this procedure (11,12). In our practice mediastinoscopy, laparoscopy and (if necessary) thoracoscopy are performed in a single OR session, and pathological analysis of the tissue samples is completed within 5 to 7 days. Once all data is obtained, disease stage is determined and a decision regarding cyto-reductive surgery is made. We argue that thorough pre-operative staging will enhance the detection of occult MPM spread to the mediastinum and peritoneum and will help improve patient selection for surgery-based MMT.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Merritt N, Blewett CJ, Miller JD, et al. Survival after conservative (palliative) management of pleural malignant mesothelioma. J Surg Oncol 2001;78:171-4. [Crossref] [PubMed]
- Vogelzang NJ, Rusthoven JJ, Symanowski J, et al. Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J Clin Oncol 2003;21:2636-44. [Crossref] [PubMed]
- Cho BC, Feld R, Leighl N, et al. A feasibility study evaluating Surgery for Mesothelioma After Radiation Therapy: the "SMART" approach for resectable malignant pleural mesothelioma. J Thorac Oncol 2014;9:397-402. [Crossref] [PubMed]
- de Perrot M, Feld R, Cho BC, et al. Trimodality therapy with induction chemotherapy followed by extrapleural pneumonectomy and adjuvant high-dose hemithoracic radiation for malignant pleural mesothelioma. J Clin Oncol 2009;27:1413-8. [Crossref] [PubMed]
- Gomez DR, Hong DS, Allen PK, et al. Patterns of failure, toxicity, and survival after extrapleural pneumonectomy and hemithoracic intensity-modulated radiation therapy for malignant pleural mesothelioma. J Thorac Oncol 2013;8:238-45. [Crossref] [PubMed]
- Sugarbaker DJ, Gill RR, Yeap BY, et al. Hyperthermic intraoperative pleural cisplatin chemotherapy extends interval to recurrence and survival among low-risk patients with malignant pleural mesothelioma undergoing surgical macroscopic complete resection. J Thorac Cardiovasc Surg 2013;145:955-63. [Crossref] [PubMed]
- Rusch VW, Giroux D, Kennedy C, et al. Initial analysis of the international association for the study of lung cancer mesothelioma database. J Thorac Oncol 2012;7:1631-9. [Crossref] [PubMed]
- Nakas A, Black E, Entwisle J, et al. Surgical assessment of malignant pleural mesothelioma: have we reached a critical stage? Eur J Cardiothorac Surg 2010;37:1457-63. [Crossref] [PubMed]
- Rusch V, Baldini EH, Bueno R, et al. The role of surgical cytoreduction in the treatment of malignant pleural mesothelioma: meeting summary of the International Mesothelioma Interest Group Congress, September 11-14, 2012, Boston, Mass. J Thorac Cardiovasc Surg 2013;145:909-10. [Crossref] [PubMed]
- van Zandwijk N, Clarke C, Henderson D, et al. Guidelines for the diagnosis and treatment of malignant pleural mesothelioma. J Thorac Dis 2013;5:E254-307. [PubMed]
- Alvarez JM, Hasani A, Segal A, et al. Bilateral thoracoscopy, mediastinoscopy and laparoscopy, in addition to CT, MRI and PET imaging, are essential to correctly stage and treat patients with mesothelioma prior to trimodality therapy. ANZ J Surg 2009;79:734-8. [Crossref] [PubMed]
- Rice DC, Erasmus JJ, Stevens CW, et al. Extended surgical staging for potentially resectable malignant pleural mesothelioma. Ann Thorac Surg 2005;80:1988-92; discussion 1992-3.
- Sørensen JB, Ravn J, Loft A, et al. Preoperative staging of mesothelioma by 18F-fluoro-2-deoxy-D-glucose positron emission tomography/computed tomography fused imaging and mediastinoscopy compared to pathological findings after extrapleural pneumonectomy. Eur J Cardiothorac Surg 2008;34:1090-6. [Crossref] [PubMed]
- Richards WG, Godleski JJ, Yeap BY, et al. Proposed adjustments to pathologic staging of epithelial malignant pleural mesothelioma based on analysis of 354 cases. Cancer 2010;116:1510-7. [Crossref] [PubMed]
- Wald O, Groth SS, Burt BM, et al. Dissection of the paratracheal plane, sampling of lymph nodes at station R4 and hemostasis are shown in the short video. Asvide 2016;3:294. Available online: http://www.asvide.com/articles/1056
Cite this article as: Wald O, Groth SS, Burt BM, Sugarbaker DJ. Role of thoracoscopy, mediastinoscopy and laparoscopy in the diagnosis and staging of malignant pleural mesothelioma. J Vis Surg 2016;2:129.