Robotic transgastric cystgastrostomy and pancreatic debridement in the management of pancreatic fluid collections following acute pancreatitis
Introduction
Acute pancreatitis results in approximately 250,000 hospitalizations yearly in the United States (1). This diagnosis is confirmed by a history of abdominal pain, elevated pancreatic enzymes, and characteristic findings on cross-sectional imaging (1,2). Approximately 5–15% of these cases develop pancreatic necrosis, which is characterized by hypoperfusion of a portion of the pancreatic parenchyma typically associated with necrosis of peripancreatic tissue (1-4). Pancreatic necrosis may not be evident on the first imaging study and can evolve over several days (4,5). Acute peripancreatic fluid collections may be visualized on imaging at the onset of pancreatitis; however, pancreatic pseudocyst or walled-off pancreatic necrosis (WOPN) both represent delayed, matured fluid collections that typically manifest at least 4 weeks after onset of symptoms. These separate entities develop by different mechanisms and are radiographically distinguishable. A pseudocyst develops from a disruption of the main pancreatic duct or an intraparenchymal ductal branch and contains amylase-rich simple fluid; characteristics include a well-defined, non-epithelialized wall and negligible to minimal solid material within the fluid collection (2). WOPN, conversely, develops from areas of pancreatic necrosis which may have been detected initially by imaging as acute necrotic collections characterized by hypoperfusion of pancreatic parenchyma (2). WOPN contains necrotic pancreatic tissue and potentially necrotic peripancreatic tissue along with a variable amount of fluid. WOPN is differentiated from an acute necrotic collection both by time from symptom onset (>4 weeks) as well as an enhancing, matured capsule of reactive tissue.
Pancreatic pseudocysts
Intervention is typically indicated for pseudocysts that fail to resolve after 6 weeks, are larger than 6 cm, and produce compressive symptoms due to their size and location; examples include gastric outlet obstruction and biliary obstruction (6,7). Pseudocysts may be treated endoscopically or surgically depending upon location (5,6). For those pseudocysts in the retrogastric or periduodenal locations, endoscopic puncture and stenting can allow for resolution via dependent internal drainage. Large series comparing surgical and endoscopic management of symptomatic pseudocysts show high rates of overall success in drainage. Endoscopically treated pseudocysts may require additional procedures to achieve complete resolution, while surgically treated pseudocysts typically require repeat procedures only for distant recurrence or bleeding due to larger anastomoses (8).
WOPN
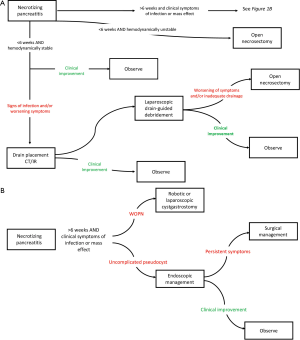
WOPN can occur in locations similar to those of pseudocysts; however, WOPN requires intervention more often for systemic symptoms or failure to thrive, recurrent fevers, or infection. Due to the inclusion of solid, necrotic material, WOPN is unlikely to completely resolve with passive drainage and requires debridement (5,8). Endoscopic instrumentation has improved to allow some debridement of the encapsulated necrosum with primary success rates of 50–80% (8-10); however, surgical debridement allows access to WOPN not directly opposed to the stomach or duodenum, with paracolic gutter extent, or with large amounts of necrotic tissue. Primary success rates for drainage of pseudocysts and WOPN in surgical series ranges from 85–100% (5,8,9). Additionally, surgical debridement of WOPN resulting from biliary pancreatitis allows for concomitant cholecystectomy. We follow an algorithmic treatment pathway for the surgical management of necrotizing pancreatitis that was developed according to our institutional experience and analysis of clinical outcomes (Figure 1).
Technique for robotic cystgastrostomy
Multiple methods for accessing pseudocysts and WOPN exist and are employed based on the location of the collection. Open, laparoscopic, and robot-assisted laparoscopic (RAL) techniques can be used; access to the necrosum can be attained via the lesser sac, through a transduodenal, endogastric, or transgastric approach or via the infracolic approach (6,7). Internal drainage procedures such as cystgastrostomy, cystduodenostomy, or cystjejunostomy with debridement are often sought to decrease the incidence of pancreaticocutaneous fistulae, as an internal enteric fistula is created to avoid transabdominal drainage (6,11).
Initial descriptions of laparoscopic transgastric cystgastrostomy were elaborated as treatment for pseudocysts (12,13); this was subsequently extended to the treatment of WOPN with inclusion of pancreatic debridement (14,15). Depending on the type of trocar used for laparoscopic transgastric pancreatic cystgastrostomy, dislodgement of trocars is feared in descriptions of the procedure. As a combination of the principles of open transgastric cystgastrostomy and the benefits of minimally invasive surgery, RAL cystgastrostomy accesses the gastric lumen via an anterior gastrotomy created with monopolar electrocautery. The superior flap of the gastrotomy can be suspended with the fourth robotic arm or sutured to the posterior surface of the anterior abdominal wall to allow freedom of all robotic arms and instruments.
The WOPN cavity has been entered classically by puncturing the point of maximum indentation into the stomach. Intraoperative ultrasound is used prior to performing a posterior gastrotomy (Figure 2 at min 2:06) to evaluate the location and extent of the necrosum relative to surrounding anatomy. After making an initial puncture into the fluid collection (Figure 2 at min 2:20), the posterior gastrotomy is extended to 5–6 cm in length with electrocautery linked to the robotic shears or the vessel sealer device. The interior of the WOPN cavity is then visualized. Necrotic pancreatic tissue is bluntly debrided using fenestrated graspers and irrigation (Figure 2 at min 2:40). Meticulous debridement of tiny pockets of necrotic tissue is not required due to the continued autodigestion of residual necrotic tissue by gastric acids facilitated by the cystgastrostomy.

Once the necrotic tissue is debrided, it is removed via a laparoscopic retrieval sac at the end of the case. Alternate reports of laparoscopic debridement describe pushing the necrotic tissue toward the pylorus for natural digestion (15). The cystgastrostomy is then sutured robotically in running fashion using absorbable barbed suture (3–0 polydioxanone V-Loc™ suture; Medtronic, Minneapolis, MN, USA) taking full-thickness bites of the cyst and gastric walls (Figure 2 at min 3:22). A stapling device is not used routinely in the robotic procedure as described in laparoscopic cystgastrostomy (14). The matured cystgastrostomy prevents separation of the posterior wall of the stomach from the WOPN cavity and is performed to decrease the incidence of anastomotic bleeding from the gastric wall and cyst wall. Prior to closing the anterior gastrotomy, a nasogastric tube is directed into the WOPN cavity for use in postoperative irrigation of the cavity. Transabdominal drains are not routinely employed postoperatively.
To close the anterior gastrotomy, multiple techniques have been described using permanent or absorbable suture and performing the closure in running or interrupted fashion as well as in one or two layers (12-15). We close the anterior gastrotomy in a single layer using 4–0 V-Loc™ suture (Medtronic) in a single layer taking full-thickness bites of the gastric wall (Figure 2 at min 4:02). Two sutures are routinely employed for closure, each starting at an apex and meeting at the midpoint of the gastrotomy, where a locking plastic clip is placed on the tails of the suture. The barbed suture is secured in place with the plastic clip and does not require a knot to be tied. We routinely reinforce the gastrotomy closure with an aerosolized fibrin sealant (TISSEEL™; Baxter Healthcare, Deerfield, Illinois, USA). At this time, other indicated procedures may be performed, such as cholecystectomy if the initial pancreatitis resulting in WOPN was biliary in origin. In the absence of other procedures or following their completion, the procedure is terminated. The abdomen is desufflated and robotic ports are removed.
Postoperative management
With the exception of nasogastric tube care, standardized enhanced recovery after surgery (ERAS) pathways are employed in the care of every patient undergoing robotic cystgastrostomy and pancreatic debridement. Specifically, deviation from ERAS pathways occurs in the use of the nasogastric tube to irrigate the retrogastric cavity with normal saline every 6 hours. The nasogastric tube is typically removed on the morning of the second postoperative day. An oral contrast swallow study is not performed to evaluate for anastomotic leak. Similarly, routine postoperative laboratory assessments are obtained on the first postoperative day but do not continue in the absence of clinical or postoperative laboratory evidence indicating repeated evaluations. Once the nasogastric tube is removed, the patients are given a noncarbonated clear liquid diet and advanced as tolerated to a regular diet. Typically, patients are discharged on postoperative day 3–5 depending on tolerance of diet and pain control. Patients are seen in clinic approximately 2 weeks after surgery and then 1 month after surgery, at which time an abdominal computed tomography (CT) scan with intravenous contrast is obtained to evaluate for resolution of the WOPN or pseudocyst.
Tips, tricks, and pitfalls
During creation of the cystgastrostomy, we demonstrate the use of the robotic shears with linked electrocautery. Using an energy device for this portion of the procedure has been described (13,15). Alternatively, though not performed during our robotic cystgastrostomy procedures, multiple firings of an endoscopic or robotic stapler could be used to create the anastomosis as in laparoscopic cystgastrostomy (8,14).
During debridement of the WOPN cavity and mobilization of the necrotic tissue, no haptic feedback exists as with laparoscopic debridement. Converting from laparoscopic to robotic debridement progressively develops a surgeon’s visual perception of tissue strain. Only after gaining sufficient robotic experience is a surgeon able to perceive how aggressively to pull and handle the tissue. We argue that the autodigestion afforded by anastomosis to the stomach allows surgeons to leave small traces of necrotic tissue in the cavity in order to prevent tearing of the cavity wall during debridement during initial experience with robotic cystgastrostomy.
Closure of the cystgastrostomy is achieved with one V-Loc™ suture initiating at each apex of the cystgastrostomy. In the video, a locking plastic clip is placed at the midpoint of the cystgastrostomy to anchor the ends of the suture. Alternatively, to avoid opening a clip applier, these barbed sutures can be continued in their respective directions beyond the midpoint to create a double-reinforced central portion of the gastrostomy closure. In this case, no clip or knot would be required.
Acknowledgements
The authors acknowledge Jennifer C. Barnes, PhD, ELS, CMPP for critical review and editing assistance.
Footnote
Conflicts of Interest: Dr. Martinie serves as a consultant and proctor for Intuitive Surgical. Dr. Iannitti serves as a consultant and proctor for Medtronic. All other authors have no conflicts of interest to declare.
References
- Lowenfels AB, Maisonneuve P, Sullivan T. The changing character of acute pancreatitis: epidemiology, etiology, and prognosis. Curr Gastroenterol Rep 2009;11:97-103. [Crossref] [PubMed]
- Banks PA, Bollen TL, Dervenis C, et al. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut 2013;62:102-11. [Crossref] [PubMed]
- Rosenberg A, Steensma EA, Napolitano LM. Necrotizing pancreatitis: new definitions and a new era in surgical management. Surg Infect (Larchmt) 2015;16:1-13. [Crossref] [PubMed]
- Stamatakos M, Stefanaki C, Kontzoglou K, et al. Walled-off pancreatic necrosis. World J Gastroenterol 2010;16:1707-12. [Crossref] [PubMed]
- Tyberg A, Karia K, Gabr M, et al. Management of pancreatic fluid collections: A comprehensive review of the literature. World J Gastroenterol 2016;22:2256-70. [PubMed]
- Aljarabah M, Ammori BJ. Laparoscopic and endoscopic approaches for drainage of pancreatic pseudocysts: a systematic review of published series. Surg Endosc 2007;21:1936-44. [Crossref] [PubMed]
- Warshaw AL, Rattner DW. Timing of surgical drainage for pancreatic pseudocyst. Clinical and chemical criteria. Ann Surg 1985;202:720-4. [Crossref] [PubMed]
- Khreiss M, Zenati M, Clifford A, et al. Cyst Gastrostomy and Necrosectomy for the Management of Sterile Walled-Off Pancreatic Necrosis: a Comparison of Minimally Invasive Surgical and Endoscopic Outcomes at a High-Volume Pancreatic Center. J Gastrointest Surg 2015;19:1441-8. [Crossref] [PubMed]
- Melman L, Azar R, Beddow K, et al. Primary and overall success rates for clinical outcomes after laparoscopic, endoscopic, and open pancreatic cystgastrostomy for pancreatic pseudocysts. Surg Endosc 2009;23:267-71. [Crossref] [PubMed]
- van Brunschot S, Fockens P, Bakker OJ, et al. Endoscopic transluminal necrosectomy in necrotising pancreatitis: a systematic review. Surg Endosc 2014;28:1425-38. [Crossref] [PubMed]
- Khaled YS, Malde DJ, Packer J, et al. Laparoscopic versus open cystgastrostomy for pancreatic pseudocysts: a case-matched comparative study. J Hepatobiliary Pancreat Sci 2014;21:818-23. [Crossref] [PubMed]
- Frantzides CT, Ludwig KA, Redlich PN. Laparoscopic management of a pancreatic pseudocyst. J Laparoendosc Surg 1994;4:55-9. [Crossref] [PubMed]
- Heniford BT, Iannitti DA, Paton BL, et al. Minilaparoscopic transgastric cystgastrostomy. Am J Surg 2006;192:248-51. [Crossref] [PubMed]
- Simo KA, Niemeyer DJ, Swan RZ, et al. Laparoscopic transgastric endolumenal cystogastrostomy and pancreatic debridement. Surg Endosc 2014;28:1465-72. [Crossref] [PubMed]
- Worhunsky DJ, Qadan M, Dua MM, et al. Laparoscopic transgastric necrosectomy for the management of pancreatic necrosis. J Am Coll Surg 2014;219:735-43. [Crossref] [PubMed]
- Kirks RC Jr, Sola R Jr, Vrochides D, et al. Robotic pancreatic cystgastrostomy with pancreatic debridement. A visualization of the robot-assisted technique of pancreatic cystgastrostomy with pancreatic debridement is provided. Asvide 2016;3:288. Available online: http://www.asvide.com/articles/1050
Cite this article as: Kirks RC Jr, Sola R Jr, Iannitti DA, Martinie JB, Vrochides D. Robotic transgastric cystgastrostomy and pancreatic debridement in the management of pancreatic fluid collections following acute pancreatitis. J Vis Surg 2016;2:127.


