Simultaneous laparoscopic distal gastrectomy (uncut Roux-en-Y anastomosis), right hemi-colectomy and radical rectectomy (Dixon) in a synchronous triple primary stomach, colon and rectal cancers patient
Introduction
Gastric cancer and colorectal cancer are common malignancies, while synchronous gastric cancer, primary colon and primary rectal cancel are extremely rare. Here we report a rare case of synchronous triple primary tumors involving gastric, colon and rectal in a 49-year-old male. Uncut Roux-en-Y anastomosis is relatively difficult procedures during distal gastrectomy, and laparoscopic radical right hemicolectomy as well as radical rectectomy (Dixon) are common procedures in colorectal surgery, while carrying out the three laparoscopic operations simultaneously has not been reported yet and the procedure is relatively complicated. Now we will describe our surgical techniques.
Methods
Ethical approval and patient consent
The present study was approved by the Ethics Committee of the First Affiliated Hospital of Nanjing Medical University (Nanjing, Jiangsu, China). Written informed consent was obtained from the present subject prior to enrollment in the present study.
Patient selection and workup
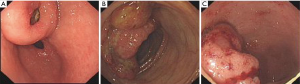
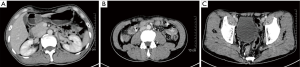
In the video (Figure 1), the patient is a 49-year-old man who was diagnosed with synchronous triple primary cancers involving gastric, colon and rectal by gastroscopy, colonoscopy and pathological examination (Figure 2). The tumor stages were evaluated to be cT1N0M0 for stomach, cT4N0M0 for colon and cT2N0M0 for rectal by CT scan pre-operation (Figure 3). Uncut Roux-en-Y anastomosis after laparoscopic distal gastrectomy with D2 lymph node dissection, laparoscopic right hemicolectomy and laparoscopic radical rectectomy for rectal cancer were intended to be carried out.

Procedure
After successful anesthesia, the patient was placed horizontal position with legs apart. At the beginning of this operation, five trocars are required. Firstly, the 10-mm trocar was placed above the umbilicus and a 30-degree laparoscope was inserted. Then other four trocars were installed under direct sight: two 5-mm trocars into the right upper and right lower abdomen, and two 12-mm trocars into the right upper and left lower abdomen.
Firstly, the peritoneal cavity and pelvic cavity were explored to rule out underlying metastasis or invasion.
Laparoscopic distal gastrectomy with D2 lymph nodes dissection procedure: after D2 lymph nodes dissection, duodenum was divided 2 cm away from pylorus using 60 mm linear stapler. The distal stomach was divided by two firings of a linear stapler, and the resected specimen was put in a plastic bag. In the same time, the jejunum 20 cm away from the Traitz ligament was labeled by silk thread.
Laparoscopic right hemicolectomy: we chose the McBurney’s point incision and lower abdomen middle incision to put in another two trocars, one 5-mm, and one 12-mm under direct vision. Then we exposed and separated the superior mesenteric vein and the ileocolic vascular, then ligated the ileocolic vascular at the root. Next we entered Toldt’s fascia to expose and prevent damage to the right gonadal vessels and ureter. Lymph nodes alone the superior mesenteric artery should be dissected and the horizontal and descending part of the duodenum should be exposed. After that we start resecting 2/3 left great omentum of the transverse colon. Ultimately, we detached the right colon and the hepatic flexure completely from the lateral peritoneal attachment.
Laparoscopic radical rectectomy: we dissected mesentery along with the right iliac artery surface up to the bifurcation, and separate mesentery along the abdominal aorta to expose inferior mesenteric artery. And then we separated the mesentery along Toldt fascia surface to the opposite side to expose and protect the left ureter. Next the sigmoid mesentery was separated at the root and lateral ligament of the rectum were divided. According to the total mesorectal excision principle, the rectum was freed along Denonvilliers fascia until at a distance of 3 cm from the distal margin of the carcinoma and divided the rectum.
Specimen extraction and anastomosis: the gastric specimen was then taken out from the abdomen through the trocar about 5 cm in size around the umbilicus. Taking out the right hemicolon through the incision around the umbilicus, divided and removed it, then an ileotransversostomy was performed by the linear stapler and strengthened by suturing. Put back the colon and took out the rectum, freed the sigmoid colon mesentery and divided the sigmoid colon about 10 cm above the tumor. The stapler head was extracorporeal placed and repositioned into the abdomen. After that, the jejunum was took out from the same incision by pulling the labeled thread and then a Braun anastomosis was performed in the proximal 10 cm and the distal 50 cm away from the Treitz ligament. Next, the afferent loop was occluded 10 cm far from the jejunojejunostomy place with linear stapler (uncut). After that, put back the small intestine into the peritoneal cavity and reestablish pneumoperitoneum. Sigmoidorectostomy was performed by stapler through anus. The last step was performing laparoscopic gastrojejunostomy at a point 2 cm distance to the blocking place.
Results
The whole operation took about 305 min with bleeding of about 300 mL. The postoperative pathologic finding showed an early gastric myxoadenocarcinoma (T1bN0M0, stage IA), colon adenocarcinoma (T4aN0M0, stage IIB), and rectal adenocarcinoma (T2N0M0, stage I). The patient discharged from hospital on the 12th day without any complication.
Discussion
Multiple primary malignant neoplasms are two or more malignancies in an individual without any relationship between the tumors. Synchronous tumors are defined as ≥2 primary tumors occurring within 6 months after diagnosing the first primary tumor. If the time interval was more than 6 months, the tumors were considered to be metachronous (2).
Both gastric and colorectal cancers are common tumor worldwide, while the morbidity of multiple synchronous primary tumors are really rare, which has been reported to vary from 0.7% to 11.7% (3). And triple synchronous primary cancers are considered has a rarer incidence. A previous study indicated that the incidence of multiple synchronous primary cancers in China is about 3.13% (4), which is similar to foreign countries. In all gastric cancer patients, Lee et al. reported that the frequency of synchronous malignant tumors is about 3.4% in 3,291 cases (5).
With the improvement of medical examination technology, more multiple synchronous primary cancers patient can be diagnosed. Nevertheless, diagnose error and misdiagnosis rate are still quite high. Therefore, when diagnosing a cancer patient, firstly, we should take synchronous primary cancers into consideration. Secondly, we should be caution when differentiating diagnosis of synchronous primary cancers from metastatic cancer, because their therapies are totally different.
Currently, there is no guideline for the treatment of multiple synchronous primary cancers. The most common treatment is surgery associated with adjuvant treatment (6). In resectable multiple synchronous primary cancers, radical surgery with or without chemotherapy is the preferred treatment (7,8). In the present synchronous triple primary stomach, colon and rectal cancers patient, we have several questions. For example, how to choose the way of operation, open or laparoscopic? If we choose laparoscopic operation on this patient, how we select the position of trocars so that we can carry out the procedure favorably and smoothly. Meanwhile, which is the optimal resection order in the present synchronous triple primary stomach, colon and rectal cancers patient? And which is the best reconstruction way for laparoscopic distal gastrectomy in this patient? Considering the relative early cStages of the three primary tumors in the present patient, we selected laparoscopic surgery and the exactly process of the operation was described detailed in the procedure part. According to our operation plan, the procedures were completed successfully, and lymph nodes dissection was sufficient, with no metastases in total 68 lymph nodes. Besides that, we made use of the essential single umbilical incision which was used to take out specimens to perform ileotransversostomy, jejunojejunostomy and occlude the afferent loop. Then laparoscopic sigmoidorectostomy and gastrojejunostomy were performed after reestablishing pneumoperitoneum. This minimally invasive operation brought the patient quickly recovery. In this case, we chose uncut Roux-en-Y reconstruction after laparoscopic distal gastrectomy with D2 lymph node dissection, because it is reported that uncut Roux-en-Y technique, would preserve unidirectional intestinal myoelectrical activity and diminish Roux Stasis Syndrome (9,10). And nine months follow-up post-operation indicates that the patient recovers well without any symptoms of Roux Stasis Syndrome.
Uncut Roux-en-Y anastomosis after laparoscopic distal gastrectomy with D2 lymph nodes dissection is a relatively complicated operation. Performing the three laparoscopic operations in one patient simultaneously is more difficult. Therefore, the laparoscopic surgery therapy for multiple primary malignant neoplasms should be performed by surgeons who are rich in experiences of laparoscopy surgical techniques.
Conclusions
In resectable multiple synchronous primary cancers, radical surgery is the preferred treatment, but weather choose the laparoscopic surgery and how to design the resection order is depending on the primary tumors’ stages and surgeons’ experience. More attempts should be done on the laparoscopic resection of multiple synchronous cancers.
Acknowledgements
Funding: “Medical Zhong Dian Ren Cai Project” of Jiangsu Province (grant No. RC2011059), the Natural Science Foundation of Jiangsu Province [grant No. BK20131447 (DA13)], “Six Ren Cai Gaofeng”.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by the Ethics Committee of the First Affiliated Hospital of Nanjing Medical University (Nanjing, Jiangsu, China). Written informed consent was obtained from the patient. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
References
- Yang L, Zhang D, Li F, et al. Uncut Roux-en-Y reconstruction after laparoscopic distal gastrectomy with D2 lymph node dissection, laparoscopic right hemicolectomy and laparoscopic radical rectectomy for rectal cancer (Dixon). Asvide 2016;3:227. Available online: http://www.asvide.com/articles/986
- Warren S, Gates O. Multiple primary malignant tumors: a survey of the literature and statistical study. Am J Cancer 1932;16:1385-414.
- Demandante CG, Troyer DA, Miles TP. Multiple primary malignant neoplasms: case report and a comprehensive review of the literature. Am J Clin Oncol 2003;26:79-83. [Crossref] [PubMed]
- Shen ZL, Wang S, Ye YJ, et al. Clinical and pathological features of synchronous double primary cancer in stomach and large intestine. Zhonghua Yi Xue Za Zhi 2008;88:162-4. [PubMed]
- Lee JH, Bae JS, Ryu KW, et al. Gastric cancer patients at high-risk of having synchronous cancer. World J Gastroenterol 2006;12:2588-92. [Crossref] [PubMed]
- Irimie A, Achimas-Cadariu P, Burz C, et al. Multiple primary malignancies--epidemiological analysis at a single tertiary institution. J Gastrointestin Liver Dis 2010;19:69-73. [PubMed]
- Kourie HR, Markoutsaki N, Roussel H, et al. Double pancreatic and gastric adenocarcinomas: a rare association. Clin Res Hepatol Gastroenterol 2013;37:e137-40. [Crossref] [PubMed]
- Wang Y, Wu XT. Stomach carcinoma presenting with a synchronous liver cancer: a case report and literature review. Case Rep Gastrointest Med 2014;2014:970293.
- Noh SM. Improvement of the Roux limb function using a new type of "uncut Roux" limb. Am J Surg 2000;180:37-40. [Crossref] [PubMed]
- Yang L, Xu ZK, Xu H, et al. Total laparoscopic uncut Roux-en-Y anastomosis in the distant gastrectomy with D1~+ or D2 dissection for gastric cancer. Chin J Prac Surg 2015;35:1099-102.
Cite this article as: Yang L, Zhang D, Li F, Ma X. Simultaneous laparoscopic distal gastrectomy (uncut Roux-en-Y anastomosis), right hemi-colectomy and radical rectectomy (Dixon) in a synchronous triple primary stomach, colon and rectal cancers patient. J Vis Surg 2016;2:101.